-
Virus Immunology (Prof. Marcus Altfeld)
Group members
Prof. Marcus Altfeld, Silke Giesemann-Jansen, Heike Hildebrandt, Dr. Maria Pujantell Graell, Dr. Kati Tillack, Dr. Yulia Zhitnyuk
Research interests
Our immune system defends us from environmental threats, such as bacterial and viral infections, and detects and removes abnormal cells that potentially lead to malignancies. Optimal immunological homeostasis is achieved when the threat (e.g. pathogen) is removed with the highest efficiency whilst avoiding collateral tissue damage for the host. Increasing scientific data, however, suggest that this immunological balance differs between women and men. It has been shown that women mount stronger immune responses against pathogens compared to men, leading to more rapid control or clearance of infections. Women also develop stronger immunity in response to vaccinations, and exhibit stronger immune responses against malignancies, including leukemia. Enhanced immune responsiveness in females, however, comes at a cost, including aggravated tissue damage, higher immune activation, persistent inflammation and significantly higher incidences of autoimmune diseases, such as autoimmune hepatitis and multiple sclerosis. However, the precise contribution of sex hormones and sex chromosome-encoded genes to sex-specific differences in immunity, and their relevance for human disease manifestations, remains insufficiently understood. The understanding of these mechanisms will pave the way for novel treatment strategies for infectious and immune-mediated diseases that take the sex of the affected individual into account.
RU 5068 "Sex differences in Immunity"
Type I IFN-mediated sex differences in immune response to HIV-1
In acute HIV-1 infection, women control viral replication better than men. In contrast, women develop increased immune activation and faster loss of CD4+ T cells during untreated chronic HIV-1 infection. Increasing data indicate that these sex differences in the manifestation of HIV-1 disease are mediated by sex-specific differences in antiviral immunity. In previous studies we demonstrated that pDCs derived from women produce more IFNα in response to HIV-1 than pDCs from men, potentially contributing to the better control of viremia in acute infection. This increased IFNα production in response to HIV-1 however also led to significantly higher immune activation and faster loss of CD4+ T cells in chronically HIV-1-infected women. Taken together these data strongly suggest that sex differences in Type I IFN production result in differences in HIV-1 disease manifestations between women and men. However, the precise molecular mechanisms underlying these sex differences in HIV-1 and other infectious diseases remain poorly understood. We hypothesize that X chromosome-encoded genes play a critical role in regulating Type I IFN responses, and that gene-dose effects resulting from escape from X chromosome inactivation (XCI) contribute to sex-specific differences in antiviral immunity, using HIV-1 as a model. We will furthermore test the hypothesis that changes in levels of sex hormones can regulate Type I IFN responses of pDCs, using longitudinal samples from a human transgender cohort. Taken together, these studies will identify critical mechanisms underlying sex-specific differences in antiviral immunity, and provide rationale for the design of interventions that take these differences between women and men into account.
Research networks
RU 5068 Research Unit 'Sex Differences in Immunity'
DZIF German Center for Infection Research
DZKJ German Center for Child and Adolescent Health
Selected publications
Schloer S, Hennesen J, Rueschpler L, Zamzamy M, Flomm F, Ip WH, Pirosu A, Dobner T, Altfeld M. (2025) The host cell factor DDX3 mediates sex dimorphism in theIFNα response of plasmacytoid dendritic cells upon TLR activation. PharmacolRes.216:107764
Padoan B, Casar C, Krause J, Schultheiss C, Baumdick ME, Niehrs A, Zecher BF, Pujantell M, Yuki Y, Martin M, Remmerswaal EBM, Dekker T, van der Bom-Baylon ND, Noble JA, Carrington M, Bemelman FJ, van Lier RAW, Binder M, Gagliani N, Bunders MJ, Altfeld M. (2024) NKp44/HLA-DP-dependent regulation of CD8 effector T cells by NK cells. Cell Rep. 43:114089
Thiele RJ, Grünhagel B, Muenchhoff M, Pujantell-Graell M, Jocham L, Düsedau A, Hennesen J, Hildebrandt H, Hagen SH, Sandfort D, Bunders MJ, Keppler OT, Hoffmann C, Altfeld M. (2024) Magnitude of Type I Interferon Responses by Plasmacytoid Dendritic Cells After TLR7 Stimulation Is Associated With Human Immunodeficiency Virus Type 1 (HIV-1) Reservoir Sizes in Cisgender Women With HIV-1 on Antiretroviral Therapy. J Infect Dis. 229:1781-1785
Pujantell M, Skenteris NT, Claussen JM, Grünhagel B, Thiele RJ, Altfeld M. (2023) Sex-dependent differences in type I IFN-induced natural killer cell activation. Front Immunol.14:1277967.
Grünhagel B, Borggrewe M, Hagen SH, Ziegler SM, Henseling F, Glau L, Thiele RJ, Pujantell M, Sivayoganathan V, Padoan B, Claussen JM, Düsedau A, Hennesen J, Bunders MJ, Bonn S, Tolosa E, Krebs CF, Dorn C, Altfeld M. (2023) Reduction of IFN-I responses by plasmacytoid dendritic cells in a longitudinal trans men cohort. iScience. ;26:108209.
Naidoo K, Altfeld M. (2023) SARS-CoV-2 exploits innate miscommunication forpersistence. Nat Immunol. 24:1974-1975
Zecher BF, Ellinghaus D, Schloer S, Niehrs A, Padoan B, Baumdick ME, Yuki Y, Martin MP, Glow D, Schröder-Schwarz J, Niersch J, Brias S, Müller LM, Habermann R, Kretschmer P, Früh T, Dänekas J, Wehmeyer MH, Poch T, Sebode M; International PSC Study Group (IPSCSG); Ellinghaus E, Degenhardt F, Körner C, Hoelzemer A, Fehse B, Oldhafer KJ, Schumacher U, Sauter G, Carrington M, Franke A, Bunders MJ, Schramm C, Altfeld M. (2024) HLA DPA1*02:01~B1*01:01 is a risk haplotype for primary sclerosing cholangitis mediating activation of NKp44+ NK cells. Gut. 73:325-337.
Baumdick ME, Niehrs A, Degenhardt F, Schwerk M, Hinrichs O, Jordan-Paiz A, Padoan B, Wegner LHM, Schloer S, Zecher BF, Malsy J, Joshi VR, Illig C, Schröder-Schwarz J, Möller KJ; Hamburg Intestinal Tissue Study Group; Martin MP, Yuki Y, Ozawa M, Sauter J, Schmidt AH, Perez D, Giannou AD, Carrington M, Davis RS, Schumacher U, Sauter G, Huber S, Puelles VG, Melling N, Franke A; International Inflammatory Bowel Disease Genetics Consortium; Altfeld M, Bunders MJ. (2023) HLA-DP on Epithelial Cells Enables Tissue Damage by NKp44+ Natural Killer Cells in Ulcerative Colitis. Gastroenterology. 165:946-962.e13.
Hagemann K, Riecken K, Jung JM, Hildebrandt H, Menzel S, Bunders MJ, Fehse B, Koch-Nolte F, Heinrich F, Peine S, Schulze Zur Wiesch J, Brehm TT, Addo MM, Lütgehetmann M, Altfeld M. (2022) Natural killer cell-mediated ADCC in SARS-CoV-2-infected individuals and vaccine recipients. Eur J Immunol. 52:1297-1307
Koyro TF, Kraus E, Lunemann S, Hölzemer A, Wulf S, Jung J, Fittje P, Henseling F, Körner C, Huber TB, Grundhoff A, Wiech T, Panzer U, Fischer N, Altfeld M. (2021) Upregulation of HLA-F expression by BK polyomavirus infection induces immune recognition by KIR3DS1-positive natural killer cells. Kidney Int. 99:1140-1148
Hagen SH, Florian Henseling, Hennesen J, Savel H, Delahaye S, Richert L, Ziegler SM, Altfeld M. (2020) Heterogeneous Escape from X Chromosome Inactivation Results in Sex Differences in Type I IFN Responses at the Single Human pDC Level. Cell Rep 33:108485
-
Infection Immunology (Prof. Hans-Willi Mittrücker)
Group members
Prof. Dr. Hans-Willi Mittrücker, Dr. Friederike Raczkowski, Melina Birus, Peter Bradtke, Twisha Kejriwal, Nameeta Jesudoss Manalan, Joanna Schmid
Research interests
The main areas of focus are:
● The function and regulation of CD4 and CD8 T cells in infections with bacterial pathogens. The analyses are carried out in infection models for Listeria monocytogenes, Staphylococcus aureus and Candida albicans.
● The role of the transcription factor Interferon Regulatory Factor 4 (IRF4) in the activation and differentiation of T cells.
● The regulation of the T cell response by the Protease A, Disintegrin and Metalloprotease 10 (ADAM10).
● Autoimmune kidney diseases and the role of T cells in the development and maintenance of immune responses against glomerular structures.
● The function and regulation of tissue-resident memory T cells in the female reproductive tract and their influence on fetal tolerance.
● The role of adenine nucleotide-activated Ca2+ channels in T cells.
Research networks
DFG: SFB 1192 „Immune-mediated glomerular diseases” (A4: “Tissue resident memory T cells in glomerulonephritis”)
DFG: SFB 1328 “Adenine Nucleotides in Immunity and Inflammation” (A03: “Adenine nucleotide-modulated T cell differentiation and effector Functions”)
DFG: SFB 1713 „Maternal Immune Activation: Causes and Consequences“ (A05: „Function and regulation of tissue resident memory T cells in the female reproductive tract: impact on maternal health during pregnancy“)
Selected publications
Asada N, Ginsberg P, Paust HJ, Song N, Riedel JH, Turner JE, Peters A, Kaffke A, Engesser J, Wang H, Zhao Y, Khatri R, Gild P, Dahlem R, Diercks BP, Das S, Ignatova Z, Huber TB, Prinz I, Gagliani N, Mittrücker H-W*, Krebs CF*, Panzer U*. The integrated stress response pathway controls cytokine production in tissue-resident memory CD4+ T cells. (2025) Nat Immunol. 26:557-566. (*shared last authorship)
Yüzen D, Urbschat C, Schepanski S, Thiele K, Arck PC, Mittrücker H-W. Pregnancy-induced transfer of pathogen-specific T cells from mother to fetus in mice. (2023) EMBO Rep. 24:e56829.
Bertram T, Reimers D, Lory NC, Schmidt C, Schmid J, C Heinig L, Bradtke P, Rattay G, Zielinski S, Hellmig M, Bartsch P, Rohde H, Nuñez S, Rosemblatt MV, Bono MR, Gagliani N, Sandrock I, Panzer U, Krebs CF, Meyer-Schwesinger C, Prinz I, Mittrücker H-W. Kidney-resident innate-like memory γδ T cells control chronic Staphylococcus aureus infection of mice. (2023) Proc Natl Acad Sci U S A. 120:e2210490120.
Harberts A, Schmidt C, Schmid J, Reimers D, Koch-Nolte F, Mittrücker H-W*, Raczkowski F*. Interferon Regulatory Factor 4 controls effector functions of CD8+ memory T cells. (2021) Proc Natl Acad Sci USA. 118:e2014553118. (*shared last authorship)
Krebs CF, Reimers D, Zhao Y, Paust HJ, Bartsch P, Nuñez S, Rosemblatt MV, Hellmig M, Kilian C, Borchers A, Enk LUB, Zinke M, Becker M, Schmid J, Klinge S, Wong MN, Puelles VG, Schmidt C, Bertram T, Stumpf N, Hoxha E, Meyer-Schwesinger C, Lindenmeyer MT, Cohen CD, Rink M, Kurts C, Franzenburg S, Koch-Nolte F, Turner JE, Riedel JH, Huber S, Gagliani N, Huber TB, Wiech T, Rohde H, Bono MR, Bonn S, Panzer U*, Mittrücker H-W*. (2020) Pathogen-induced tissue-resident memory TH17 (TRM17) cells amplify autoimmune kidney disease. Sci Immunol. 5:eaba4163. (*shared last authorship)
Collaboration partners
University Medical Center Hamburg-Eppendorf (UKE)
Marcus Altfeld, Friedrich Koch-Nolte, Felix Stahl, Eva Tolosa Institute for Immunology Ulf Panzer, Christian Krebs, Ulrich Wenzel III. Medical Clinic and Polyclinic Samuel Huber, Nicola Gagliani I. Medical Clinic and Polyclinic Petra Arck Experimental feto-maternal medicine Torsten Wiech Institute of Pathology Andreas Guse, Björn-Philipp Dierks Institute for Biochemistry and Molecular Cell Biology Björn Rissiek, Tim Magnus Department of Neurology Cathy Meyer-Schwesinger Institute of Cellular and Integrative Physiology
external partners
Minka Breloer, Hanna Lotter Bernhard Nocht Institute for Tropical Medicine, Hamburg Magdalena Huber Philipps University of Marburg Stefan Rose-John Christian-Albrechts University of Kiel Santina Bruzzone University of Genova
-
Molecular Immunology (Prof. Friedrich Koch-Nolte)
The Molecular Immunology group studies membrane-bound receptors and enzymes of immune and cancer cells and develops nanobodies and antibodies against these targets.
Group members
Prof. Friedrich Koch-Nolte, Dr. rer. nat. Anna Marei Mann, Dr. rer. nat. Julia Hambach, Anya Duttmann, Josephine Gebhardt, Saruul Jargalsaikhan, Luca Pape, Nathalie Richter, Waldemar Schäfer, Fabienne Seyfried, Natalie Tode
Research networks
DFG: SFB 1192 „Immune-mediated glomerular diseases” (B05: Nanobody-based treatment strategies in glomerulonephritis, Nicola Wanner, Friedrich Nolte)
DFG: SFB 1328 “Adenine Nucleotides in Immunity and Inflammation” (A10: Role of adenine nucleotides and purinergic receptors in adipose tissue, Jörg Heeren, Friedrich Nolte; Z02: Antibodies and nanobodies for adenine nucleotide research Friedrich Haag, Friedrich Nolte)
DFG: FOR 2879 "ImmunoStroke: From Immune Cells to Stroke Recovery" (A1: Role of ATP and NAD as DAMP signals in stroke recovery and as therapy targets, Tim Magnus, Friedrich Nolte)
DFG/ANR: Evaluation of P2X7 as a therapeutic target in autoimmune encephalomyelitis and in tumor immunity (Sahil Adriouch, Univ. Rouen, Friedrich Nolte)
DFG: Engineering the avidity of nanobody-based bispecific diabodies and heavy chain antibodies to specifically target myeloma cells that co-express two membrane proteins (Peter Bannas, Radiologie, Friedrich Nolte)
BMBF COMMUTE Combinatorial and multidisciplinary targeting of effective gene therapy vectors (P5 Targeting glioblastoma and muscle dystrophy with Nanobody-displaying AAV, Friedrich Nolte)
Short CV Prof. Friedrich Koch-Nolte
Deputy Director (since 1997)
Phone: +49 (0) 40 7410 - 53612
Mail: nolte@uke.deUniversity Degrees
Biology, Wesleyan University 1976 (BA)
Medicine, University of Tübingen, 1983 (MD)
Molceular Biology, University of Hamburg, 1986 (Diploma)
Professor associée at the University of Rouen, France (2006-2007)
Visiting scientist at the University of California, San Francisco, CA, USA (1994)
Visiting scientist at the DNAX Research Institute of Molecular Biology, Palo Alto, CA, USA (1997)
Visiting scientist at The Jackson Lab, Bar Harbor, ME, USA (1991, 1999)
Research award of the German Cancer Society (2019)Mentorship Award of the Simon-Claussen Foundation (2009)
Research Award of the Werner Otto Foundation (1997)
Research Award of the Martini Foundation (1991)
Member of the
Deutsche Gesellschaft für Immunologie (DGfI)
Deutsche Gesellschaft für Biochemie und Molekularbiologie (GBM)
Deutsche Gesellschaft für Zellbiologie (DGZ)Collaboration partners
University Medical Center Hamburg-Eppendorf (UKE)
Eva Tolosa, Hans-Willi Mittrücker, Felix Stahl Institute for Immunology Andreas Guse, Jörg Heeren, Ralf Fliegert Institute for Biochemistry and Molecular Cell Biology Peter Bannas Department of Diagnostic and Interventional Radiology and Nuclear Medicine Tim Magnus, Björn Rissiek Department of Neurology Cathy Meyer-Schwesinger Institute of Cellular and Integrative Physiology Nicola Wanner, Nicola Tomas, Elion Hoxha, Rolf Stahl, Ulf Panzer III. Medical Clinic and Polyclinic Boris Fehse, Kristoffer Rieken Department of Stem Cell Transplantation Irm Hermanns Borgmeyer Center for Molecular Neurobiology Katja Weisel, Walter Fiedler II. Medical Clinic and Polyclinic Thomas Braulke Institute of Osteology and Biomechanics
external partners
Sahil Adriouch University of Rouen Normandy Michael Hottiger University of Zurich Pablo Pelegrin University of Murcia Mascha Binder University of Halle Martin Trepel University of Augsburg Dirk Grimm University of Heidelberg Annette Nicke Ludwig Maximilian University of Munich Carsten Watzl Technical University of Dortmund Florian Schmidt University of Bonn Francesco Di Virgilio, Elena Adinolfi University of Ferrara Santina Bruzzone, Paolo Malatesta University of Genoa Jan van Lier University of Amsterdam Jaime Sancho University of Granada Pablo Engel University of Barcelona Ursula Dietrich Georg-Speyer-Haus William Heath University of Melbourne William Petri University of Virginia Steven Mansoor University of Oregon Hidde Ploegh Boston Children's Hospital
Research interests
The focus of our laboratory is on the molecular characterization of lymphocyte membrane proteins, in particular receptors and enzymes involved in signaling by extracellular nucleotides. We generate monoclonal and single domain antibodies as new research and therapeutic tools. We are interested in ADP-ribosylation as a pathogenic mechanism of bacterial toxins and as a reversible posttranslational modification regulating protein functions. Using genetic immunization and antibody engineering, we strive to develop new tools for combating infections and for treating diseases of the immune system.
1. Lymphocyte membrane proteins: enzymes and receptors involved in signaling by extracellular NAD and ATP
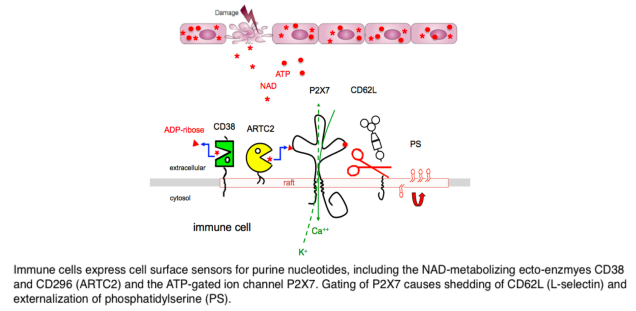
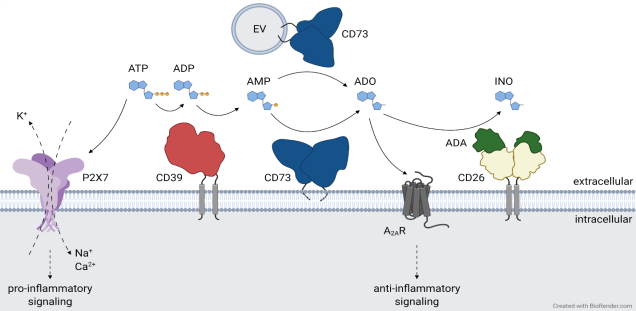
Membrane proteins mediate the communication of cells with their environment. They function as receptors for soluble ligands and counter-receptors on other cells, as ion channels, nutrient transporters, and enzymes. The nucleotides NAD and ATP are key metabolites of energy metabolism found in cells from all kingdoms of life. The cell membrane is impermeable to these nucleotides, but they can exit cells via channels or pores gated by mechanical and/or electrical stimuli. During infection and inflammation injured cells release NAD and ATP through the damaged cell membrane. Extracellular NAD and ATP alert cells of the immune system to sites of tissue damage.Cells of the immune system are equipped with a variety of sensors for these nucleotides (Fig. 1), including ligand-gated ion channels (P2X purine receptors) and nucleotide-metabolizing ecto-enzymes (CD38, CD296). CD296 (ARTs) functions as NAD-sensors and relay information about the levels of extracellular NAD into ADP-ribosylation of cell surface and secreted proteins. The NAD-hydrolyzing ecto-enzyme CD38 restricts the intensity and duration of NAD-signaling in the extracellular space by hydrolyzing NAD. Opening of the P2X7 ion channel is induced by binding of the soluble ligand ATP, or by NAD-dependent ADP-ribosylation. Passage of ions through P2X7 (calcium into the cell, potassium out of the cell) triggers a cascade of downstream events, including protease activation (caspases and ADAMs), externalization of phosphatidylserine (PS), activation of the inflammasome and cell death. Mice that cannot express the mentioned purine receptors or ecto-enzymes show impaired immune responses. The receptors and enzymes of purinergic signaling, thus, present potential targets for new anti-inflammatory or immune-stimulating drugs. In mouse models, activating the ARTC2 > P2X7 axis can enhance anti-tumor responses, while blocking this axis can reduce inflammation in autoimmune diseases.
2. Antibody engineering: genetic immunization, single domain antibodies
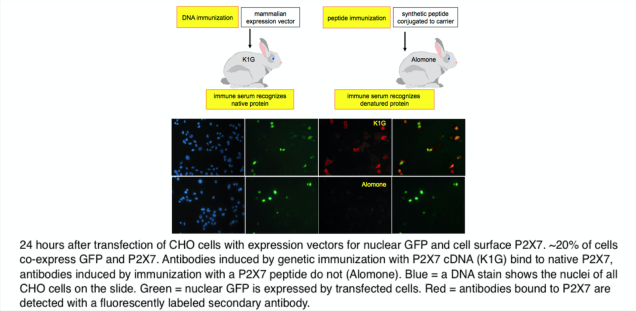
Antibodies are important tools of research, diagnosis and therapy. Antibodies are raised by immunizing experimental animals with proteins, synthetic peptides, or DNA. In the case of DNA immunizations, the protein encoded by the DNA is produced in its native conformation by the cells of the immunized animal and the induced immune response yields antibodies directed against proteins in native conformation (ADAPINCs) (Fig. 2). Such antibodies are useful in many applications were anti-peptide antibodies fail, e.g. affinity purification, flow cytometry, ELISA, FACS, and functional studies. cDNA immunization is offered as a service via the antibody core facility of the UKE.
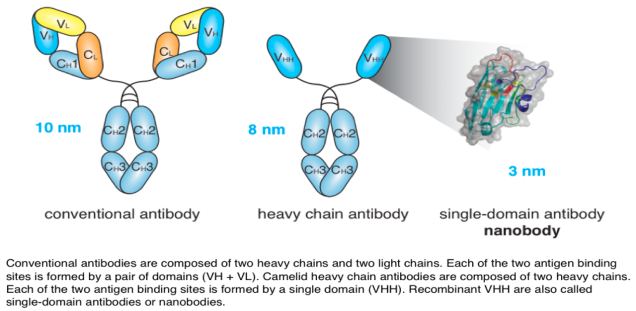
Llamas produce unusual antibodies composed only of heavy chains. Their antigen-binding domain (VHH) is readily produced as a soluble recombinant protein, also designated nanobody or single domain antibody (sdAb) (Fig. 3). Nanobodies can form finger like extrusions that block clefts on protein surfaces, such as the active site crevice of enzymes, the ligand binding domain of a receptor, and the receptor binding domain of a virus. Nanobodies have great potential as therapeutics and as imaging agents.
We have generated enzyme-blocking nanobodies from llamas immunized with different ARTs: the SpvB Salmonella toxin, the binary clostridium difficile toxin CDTa, and the T cell ecto-enzyme ARTC2 (CD296). These nanobodies protect cells from the cytotoxic effects of SpvB, CDT, and ARTC2. In case of ARTC2, the nanobodies effectively block ARTC2 on the cell surface of regulatory T cells and iNKT cells within 10 minutes after intravenous injection. These nanobodies provide an important tool for protecting these regulatory cells from death induced by NAD released during tissue preparation.
We have also generated nanobodies against CD38, the major NAD-hydrolyzing ecto-enzyme. CD38 is emerging as a therapeutic target in multiple myeloma and other hematopoertic malignancies. Some of our nanobodies outperform the recently licensed CD38-specific monoclonal antibody Daratumumab (Darzalex) in cytotoxicity vs. hematopoetic cancer cell lines.
In cooperation with Ablynx, a Belgian company devoted to developing nanobodies for human therapies, we have generated nanobodies that block or enhance gating of the P2X7 ion channel. The P2X7-blocking nanobodies ameliorated inflammation in mouse models of glomerulonephritis and allergic dermatitis. In endotoxin-treated human blood cells, they effectively blocked LPS/ATP-induced release of the potent pro-inflammatory cytokine IL-1ß.
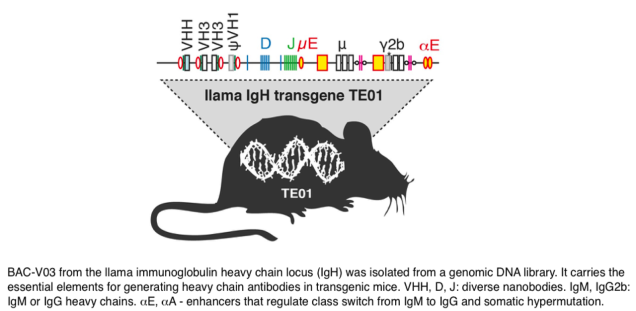
In order to facilitate the generation of new nanobodies, we have cloned the nanobody-encoding IgH locus from llama and successfully transferred an engineered version of this locus to transgenic mice (Fig. 4). Upon immunization, these mice produce nanobody-based heavy chain antibodies that undergo somatic hypermutation and class switch recombination. This novel platform greatly expands our capacity to generate functional nanobodies against interesting targets.3. Posttranslational modifications: ADP-ribosylation, glycosylation, lipid anchors
The function of proteins can be regulated via the attachment of chemical moieties. These enzyme-catalyzed modifications, coined posttranslational modifications (PTMs), include phosphorylation, glycosylation, ADP-ribosylation, and the attachment of lipid anchors.
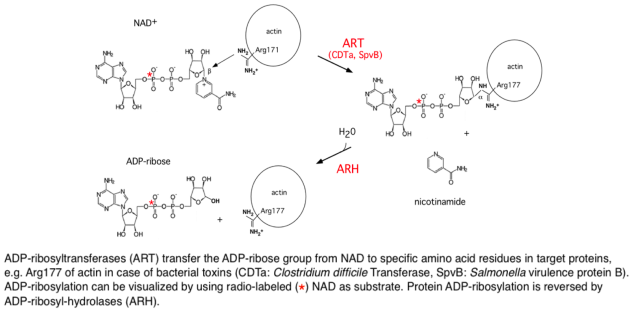
ADP-ribosylation is a reversible PTM, in which ADP-ribosyltransferases (ARTs) transfer the ADP-ribose moiety from NAD onto a specific amino acid side chain in a target protein and ADP-ribosylhydrolases (ARHs) remove the ADP-ribose group (Fig. 5). We have determined the 3D structures of a prototype ART and a prototype ARH. The 3D structure of rat ARTC2 resembles a pacman with a wide-open mouth crunching on the substrate NAD. The 3D structure of human ARH3 resembles a pumpkin in which four alpha helices coordinate two magnesium ions at the bottom of the active site crevice.ADP-ribosylation is used by pathogenic toxins such as Diphtheria, Pertusssis and Clostridial toxins to modulate host protein functions. Toxin-related ARTs are expressed by cells of the immune system. ADP-ribosylation of membrane proteins can be monitored using labeled analogues of NAD. Using 32P-NAD, ADP-ribosylation results in radiolabeling of the target protein. Using etheno-NAD, ADP-ribosylation of target proteins can be detected with a monoclonal antibody directed against etheno-adenosine. This 1G4 antibody can be used to sort cells on the basis of cell surface ART-activity and to purify etheno-ADP-ribosylated proteins.
ARTC2 itself is posttranslationally modified, e.g. by attachment of a GPI lipid anchor (Fig. 1 above). The GPI-anchor targets ARTC2 to lipid rafts, specialized regions of the cell membrane that play an important role in signal transduction, e.g. during activation of T cells by antigen presenting cells. The association of ARTC2 with lipid rafts focuses ARTC2 onto specific target proteins and thereby may regulate the signaling function of raft-associated proteins. ADP-ribosylation of the P2X7 ion channel on arginine residue 125 activates P2X7 to form a non-selective ion-channel, permitting calcium ions to enter the cell and potassium ions to exit the cell. This induces dramatic alterations of the cell membrane, including the externalization of PS, shedding of L-selectin, and formation of membrane blebs.
Selected publications
reviews
Eggers M, Rühl F, Haag F, Koch-Nolte F. (2021) Nanobodies as probes to investigate purinergic signaling. Biochem Pharmacol. 87:114394. review PMID: 33388283.
Linden J, Koch-Nolte F, Dahl G. (2019) Purine Release, Metabolism, and Signaling in the Inflammatory Response. Annu Rev Immunol. 37:325-347. review. PMID: 30676821
Stortelers C, Pinto-Espinoza C, Van Hoorick D, Koch-Nolte F. (2018) Modulating ion channel function with antibodies and nanobodies. Curr Opin Immunol. 52:18-26. Review PMID: 29579624
Bannas P, Hambach J, Koch-Nolte F. (2017) Nanobodies and Nanobody-Based Human Heavy Chain Antibodies As Antitumor Therapeutics. Front Immunol. 8:1603. Review PMID: 29213270
Wesolowski J, Alzogaray V, Reyelt J, Unger M, Juarez K, Urrutia M, Cauerhff A, Danquah W, Rissiek B, Scheuplein F, Schwarz N, Adriouch S, Boyer O, Seman M, Licea A, Serreze DV, Goldbaum FA, Haag F, Koch-Nolte F. (2009) Single domain antibodies: promising experimental and therapeutic tools in infection and immunity. Med Microbiol Immunol. 198:157-74. Review PMID: 19529959
Koch-Nolte F, Kernstock S, Mueller-Dieckmann C, Weiss MS, Haag F. (2008) Mammalian ADP-ribosyltransferases and ADP-ribosylhydrolases. Front Biosci. 13:6716-29. PMID: 18508690
research reports
Hambach J, Stähler T, Eden T, Wendt D, Tode N, Haag F, Tolosa E, Altfeld M, Fathi A, Dahlke C, Addo MM, Menzel S, Koch-Nolte F. (2021) A simple, sensitive, and low-cost FACS assay for detecting antibodies against the native SARS-CoV-2 spike protein. Immun Inflamm Dis. 9:905-917. PMID: 33979020.
Schriewer L, Schütze K, Petry K, Hambach J, Fumey W, Koenigsdorf J, Baum N, Menzel S, Rissiek B, Riecken K, Fehse B, Röckendorf JL, Schmid J, Albrecht B, Pinnschmidt H, Ayuk F, Kröger N, Binder M, Schuch G, Hansen T, Haag F, Adam G, Koch-Nolte F, Bannas P. (2020) Nanobody-based CD38-specific heavy chain antibodies induce killing of multiple myeloma and other hematological malignancies. Theranostics. 10:2645-2658. PMID: 32194826
Eichhoff AM, Börner K, Albrecht B, Schäfer W, Baum N, Haag F, Körbelin J, Trepel M, Braren I, Grimm D, Adriouch A, Koch-Nolte F. (2019) Nanobody-enhanced targeting of AAV gene therapy vectors Mol Ther Methods Clin Dev. 15:211-220. PMID: 31687421
Eden T, Menzel S, Wesolowski J, Bergmann P, Nissen M, Dubberke G, Seyfried F, Albrecht B, Haag F, Koch-Nolte F. (2018) A cDNA Immunization Strategy to Generate Nanobodies against Membrane Proteins in Native Conformation. Front Immunol. 8:1989. PMID: 29410663
Danquah W, Meyer-Schwesinger C, Rissiek B, Pinto C, Serracant-Prat A, Amadi M, Iacenda D, Knop JH, Hammel A, Bergmann P, Schwarz N, Assunção J, Rotthier W, Haag F, Tolosa E, Bannas P, Boué-Grabot E, Magnus T, Laeremans T, Stortelers C, Koch-Nolte F. (2016) Nanobodies that block gating of the P2X7 ion channel ameliorate inflammation. Sci Transl Med. 8:366ra162. PMID: 27881823
Adriouch S, Bannas P, Schwarz N, Fliegert R, Guse AH, Seman M, Haag F, Koch-Nolte F. (2008) ADP-ribosylation at R125 gates the P2X7 ion channel by presenting a covalent ligand to its nucleotide binding site. FASEB J. 22:861-9. PMID: 17928361
Koch-Nolte F, Reyelt R, Schössow B, Schwarz N, Scheuplein F, Rothenburg S, Haag F, Alzogaray V, Cauerhff A, Goldbaum F. (2007) Single domain antibodies from llama effectively and specifically block T cell ecto-ADP-ribosyltransferase ART2.2 in vivo. FASEB J. 21:3490-8. PMID: 17575259
Mueller-Dieckmann C, Kernstock S, Lisurek M, von Kries JP, Haag F, Weiss MS, Koch-Nolte F. (2006) The structure of human ADP-ribosylhydrolase 3 (ARH3) provides insights into the reversibility of protein ADP-ribosylation. Proc Natl Acad Sci U S A. 103:15026-31. PMID: 17015823
Otto H, Reche PA, Bazan F, Dittmar K, Haag F, Koch-Nolte F. (2005) In silico characterization of the family of PARP-like poly(ADP-ribosyl)transferases (pARTs). BMC Genomics. 6:139. PMID: 16202152
Bannas P, Adriouch S, Kahl S, Braasch F, Haag F, Koch-Nolte F. (2005) Activity and specifity of toxin-related mouse T cell ecto-ADP-ribosyltransferase ART2.2 depends on it association with lipid rafts. Blood. 105:3663-70. PMID: 15657180
Seman M, Adriouch S, Scheuplein F, Krebs C, Freese D, Glowacki G, Deterre P, Haag F, Koch-Nolte F. (2003) NAD-induced T cell death: ADP-ribosylation of cell surface proteins by ART2 activates the cytolytic P2X7 purinoceptor. Immunity. 19:571-82. PMID: 14563321
Otto H, Tezcan-Merdol D, Girisch R, Haag F, Rhen M, Koch-Nolte F. (2000) The spvB gene-product of the Salmonella enterica virulence plasmid is a mono(ADP-ribosyl)transferase. Mol Microbiol. 37:1106-15. PMID: 10972829
Koch-Nolte F, Petersen D, Balasubramanian S, Haag F, Kahlke D, Willer T, Kastelein R, Bazan F, Thiele HG. (1996) Mouse T cell membrane proteins Rt6-1 and Rt6-2 are arginine/protein mono(ADPribosyl)transferases and share secondary structure motifs with ADP-ribosylating bacterial toxins. J Biol Chem. 271:7686-93. PMID: 8631807
Koch F, Haag F, Kashan A, Thiele HG. (1990) Primary structure of rat RT6.2, a nonglycosylated phosphatidylinositol-linked surface marker of postthymic T cells. Proc Natl Acad Sci U S A. 87:964-7. PMID: 2300588
Koch F, Thiele HG, Low MG. (1986) Release of the rat T cell alloantigen RT-6.2 from cell membranes by phosphatidylinositol-specific phospholipase C. J Exp Med. 164:1338-43. PMID: 3489808
-
Medical Immunology (Prof. Felix Stahl)
Group members
Prof. Felix Stahl, Dr. Luis Fonseca Brito, Lama Al Jurdi, Emily Kerth, Nan Ma, Dorottya Schöffer, Victoria Tennert
Research interests
Viral infections trigger complex, well-orchestrated host responses involving both humoral and cellular immunity. Among these viruses, herpesviruses are large, highly adapted pathogens that have co-evolved with their hosts to counteract immune defenses. Cytomegalovirus (CMV), a prototype member of the Betaherpesvirinae family, encodes multiple molecules that enable it to evade antiviral immunity. CMV establishes a chronic, persistent infection and is typically considered an opportunistic pathogen; however, disruptions in host immunity can lead to increased viral replication and the manifestation of CMV-related diseases, particularly in immunocompromised individuals. Notably, congenital CMV infection is the leading cause of permanent disabilities in newborns, yet no vaccine exists, and current antiviral therapies lack efficacy.
We study the interaction between host immune cells and viral infections in clinically relevant contexts, such as early life infection, utilizing state-of-the-art methods and model systems. We focus on understanding how leukocytes mediate their antiviral effects at the site of infection and aim to overcome the challenges associated with controlling viral infections.
Research networks
DFG: FOR 5068 "Sex Differences in Immunity"
DFG: CRC 1713 "Maternal Immune Activation: Causes and Consequences"
Selected publications
Fonseca Brito L, Ostermann E, Perez A, Tödter S, Virdi S, Indenbirken D, Glau L, Gieras A, Brixel R, Arens R, Grundhoff A, Arck P, Diemert A, Tolosa E, Brune W, Stahl FR. (2024) Limited protection against early-life cytomegalovirus infection results from deficiency of cytotoxic CD8 T cells. bioRxiv. 2024.07.10.602923.
Fonseca Brito L, Todter S, Kottlau J, Cermann K, Spier A, Petersen E, Schafer I, Twerenbold R, Aepfelbacher M, Lutgehetmann M, Stahl FR. Performance of an interferon-gamma release assay-based test for cell-mediated immunity to SARS-CoV-2. (2023) Front Immunol. 14:1069968.
Fonseca Brito L, Brune W, Stahl FR. Cytomegalovirus (CMV) Pneumonitis: Cell Tropism, Inflammation, and Immunity. (2019) Int J Mol Sci. 20(16).
Stahl FR, Jung R, Jazbutyte V, Ostermann E, Todter S, Brixel R, Kemmer A, Halle S, Rose-John S, Messerle M, Arck PC, Brune W, Renne T. (2018) Laboratory diagnostics of murine blood for detection of mouse cytomegalovirus (MCMV)-induced hepatitis. Sci Rep. 8(1):14823.
Lueder Y, Heller K, Ritter C, Keyser KA, Wagner K, Liu X, Messerle M, Stahl FR#, Halle S#, Forster R#. Control of primary mouse cytomegalovirus infection in lung nodular inflammatory foci by cooperation of interferon-gamma expressing CD4 and CD8 T cells. (2018) PLoS Pathog. 14(8):e1007252.
Halle S, Keyser KA, Stahl FR, Busche A, Marquardt A, Zheng X, Galla M, Heissmeyer V, Heller K, Boelter J, Wagner K, Bischoff Y, Martens R, Braun A, Werth K, Uvarovskii A, Kempf H, Meyer-Hermann M, Arens R, Kremer M, Sutter G, Messerle M, Forster R. (2016) In Vivo Killing Capacity of Cytotoxic T Cells Is Limited and Involves Dynamic Interactions and T Cell Cooperativity. Immunity. 44(2):233-45.
Stahl FR#, Keyser KA#, Heller K, Bischoff Y, Halle S, Wagner K, Messerle M, Forster R. Mck2-dependent infection of alveolar macrophages promotes replication of MCMV in nodular inflammatory foci of the neonatal lung. (2015) Mucosal Immunol. 8(1):57-67.
Stahl FR, Heller K, Halle S, Keyser KA, Busche A, Marquardt A, Wagner K, Boelter J, Bischoff Y, Kremmer E, Arens R, Messerle M, Forster R. (2013) Nodular inflammatory foci are sites of T cell priming and control of murine cytomegalovirus infection in the neonatal lung. PLoS Pathog.9(12):e1003828.
#shared authorships
-
Immune Regulation (Prof. Eva Tolosa)
Group members
Prof. Eva Tolosa, Finja Beer, Barsa Dhal, Carolin Diener, Laura Glau, Romy Hackbusch, Zoe-Isabella Junginger, Manuela Kolster, Jonathan May, Du-Hanh Nguyen, Friederike Thiele, Dr. Kati Tillack, Timon Warschau, Dr. Riekje Winzer
Research interests
Adenine nucleotides and immune regulation
Expression, function and regulation of purinergic receptors and adenine nucleotide-metabolizing ectoenzymes.
Adenine nucleotides play a key role in the modulation of the immune response. In our research group, we investigate the release of adenine nucleotides during inflammatory responses, the activation of purinergic receptors, and the degradation of adenine nucleotides by ectoenzymes.
The concentration of extracellular adenosine triphosphate (ATP) is low under physiological conditions, but increases significantly during inflammatory responses or cellular stress. Higher concentrations of ATP are also present in some tissues, such as the intestine. Extracellular ATP serves as a ligand for P2 receptors, and stimulation of P2X7 receptors on immune cells promotes the release of pro-inflammatory cytokines. The coordinated action of the ectoenzymes CD39 and CD73 converts ATP into adenosine (ADO), providing a dual function, which is the elimination of the danger signal ATP and the generation of the anti-inflammatory mediator ADO. This illustrates a mechanism to control the outcome of an immune response.
In some diseases, such as tumor diseases, it is therapeutically beneficial to enhance the immune response, whereas other disease conditions, such as autoimmune diseases, require immunosuppressive therapy. Modulating the ATP-ADO-axis by regulation of ectoenzymes and purinergic receptors thus represents an interesting research target for the first step in developing immunomodulatory therapies. Using inhibitors and knock-out systems, we want to modulate the cell-specific influence of these ectoenzymes and receptors and thus characterize potential targets for the regulation of immune responses.
Current projects in the lab related to adenine nucleotide signaling are:
Dissecting the role of ATP and adenosine receptor signaling forimmune adaptation of intestinal Th17 cells, PI: Prof. Eva Tolosa, Prof. Nicola Gagliani; CRC 1328 subproject A14.
In inflamed tissue, Th1 and Th17 cells mediate tissue-damaging processes by secreting pro-inflammatory cytokines. Some of these effector cells possess a certain degree of plasticity and can adopt an anti-inflammatory phenotype (a process referred to as immune adaptation), contributing to the resolution of inflammation. Our hypothesis is that the hydrolysis of extracellular ATP to ADO by Th1 and Th17 cells, and ADO receptor-mediated signaling pathways promote their conversion into regulatory cells. To decipher the role of ectonucleotidases CD39 and CD73 in controlling the availability of ADO, and the role of ADO receptor signaling in immune adaptation, we will perform experiments in vitro using human Th1 and Th17 cells, and, in collaboration with Prof. Nicola Gagliani, use cytokine reporter mice in the context of intestinal inflammation.
Here you can find more information about the CRC 1328
Generation of adenosine and its role in post-stroke immune suppression, PIs: Prof. Eva Tolosa (Immunology, UKE Hamburg), Prof. Götz Thomalla (Neurology, UKE Hamburg), and Profs. Luisa Klotz and Heinz Wiendl (Neurology, Uni. Muenster); RU 2879 subproject C1
Ischemic stroke induces a rapid immunologic response characterized by the invasion of leukocytes from the periphery into the ischemically damaged region. At the same time, clinically relevant immunosuppression can be observed in stroke patients. The aim of this project is to elucidate the dynamics of stroke-related changes in the immune signature and functional properties of peripheral blood immune cells, with a focus on purinergic signaling as potential mechanism underlying stroke-induced immune suppression. Changes in the human immunome will be evaluated in relation to stroke subtype, characteristics of ischemic stroke lesions and disease outcome, with the aim of an early identification and specific treatment of risk-patients.
Here you can find more information about the RU 2879
Thymus function in health and disease
The thymus, a primary lymphoid organ, plays a crucial role in T cell development. Tightly controlled selection processes ensure the establishment of a functional but self-tolerant immune response against pathogens or tumors, without leading to autoimmunity. Around birth, thymic function is at its peak and therefore, events affecting the thymus during this sensitive period of time may have serious consequences for the child’s immunity.
In our lab, we explore a range of early life factors that lead to a decline or even loss of thymic function. Causes can be various and range from medical interventions (e.g. prenatal steroids, early life thymectomy) to inborn disorders (e.g. Down syndrome). The consequences may persist well beyond the first months of life and lead to an impaired immunity (e.g. development of chronic or autoimmune diseases).
Consequences of congenital heart disease and early thymectomy on immune development PI: Dr. Anna Gieras, supported by the German Heart Foundation
Early life medical interventions can be detrimental to thymic development and need to be cautiously evaluated to prevent unwanted adverse effects. We are investigating the impact of early life thymectomy on thymic function and development of the adaptive immune system.
Mechanistic insights into thymic function and immune dysfunction in Down Syndrome PI: Dr. Anna Gieras, supported by the Werner-Otto Stiftung
We study the impact of congenital diseases on poor thymic function with a major focus on patients with Down syndrome. These patients exhibit an altered peripheral immune system but the nature of the defect causing changes in the peripheral T cell pool are not yet known and a detailed analysis of thymic function is missing. Our preliminary data show a defective development of immature thymocytes (ex vivo and in vitro) and we are currently investigating underlying molecular mechanisms.
In vitro studies of thymic function in humans are challenging, mainly due to limitations in the availability of thymic tissue. In order to mimic the thymic environment in vitro we have established 3D human thymic organoids to assess and manipulate the development of T lymphocytes from patient-derived progenitor cells.
Immune phenotyping
High resolution multiparameter flow cytometry for profiling the immune system in health and disease
Profiling the immune system is to get a snapshot of an individual’s immune health, and allows the identification of immune traits associated to a disease condition, genetic defect, ageing, etc. While immune cells in diseased organs can show substantial differences compared to their physiological state, changes in peripheral blood are often very subtle. Therefore, deep immune phenotyping including assessment of function and project-specific bioinformatics analysis are necessary to detect disease- or group-associated traits ( RU 2879, C1 ). In our lab we have developed panels that are specific to address the immune status in relation to age, with specific panels developed for cord blood analysis; response to vaccination; for comparison of peripheral blood to organ- and tumor-infiltrating cells, for assessment of disease-related immune traits, etc.. We use a flow bioinformatics pipeline for quick evaluation of changes in the immune status of individuals, and for unbiased analysis of raw data for the quantification of differentially abundant clusters in pre-defined groups of patients or cellular compartments.
Research networks
DFG: CRC 1328 Adenine nucleotides in immunity and inflammation
DFG: RU 2879 ImmunoStroke
DFG: KFO 5029 ProBone
Selected publications
Bremer S#, Boxnick A#, Glau L, Biermann D, Joosse S, Thiele F, Billeb E, May J, Kolster M, Hackbusch R, Fortmann M, Kozlik-Feldmann R, Hübler M, Tolosa E, Sachweh J, Gieras A (2024) Thymic Atrophy and Immune Dysregulation in Infants with Complex Congenital Heart Disease. J Clinical Immunology (2024) 44:69
Winzer R, Serracant-Prat A, Brock V, Pinto-Espinoza C, Rissiek B, Amadi M, Eich N, Rissiek A, Schneider E, Magnus T, Guse A, Diercks B, Koch-Nolte F, Tolosa E (2022) P2X7 is expressed on human innate-like T lymphocytes and mediates susceptibility to ATP-induced cell death EEUR J IMMUNOL. 2022;52(11):1805-1818.
Schneider E#, Winzer R#, Rissiek A, .., Magnus T, Fliegert R, Müller CE, Gagliani N, Tolosa E (2021) CD73-mediated adenosine production by CD8 T cell-derived extracellular vesicles constitutes an intrinsic mechanism of immune suppression in humans. Nat Commun. 8;12:5911.
Poch T, Krause J, Casar C, Liwinski T, Glau L, …Huber S, Tolosa E#, Gagliani N#, Schramm C# (2021) Single-cell atlas of hepatic T cells reveals expansion of liver-resident naive-like CD4+ T cells in primary sclerosing cholangitis. J Hepatol. S0168-8278(21)00219-1 3.
Bremer SJ, Glau L, Gehbauer C, Boxnick A, Biermann D, Sachweh JS, Tolosa E, Gieras A. (2021) OMIP 073: Analysis of human thymocyte development with a 14-color flow cytometry panel. Cytometry A. 99(9):875-879.
Lessel D, Gehbauer C, Bramswig NC, Schluth-Bolard C, Venkataramanappa S, van Gassen KLI,… Agrawal PB, Britsch S, Tolosa E#, Kubisch C# (2018) BCL11B mutations in patients affected by a neurodevelopmental disorder with reduced type 2 innate lymphoid cells. Brain; 141(8):2299-311
Mohme M, Schliffke S, Maire CL, Runger A, Glau L, … Westphal M, Binder M#, Tolosa E#, Lamszus K# (2018) Immunophenotyping of Newly Diagnosed and Recurrent Glioblastoma Defines Distinct Immune Exhaustion Profiles in Peripheral and Tumor-infiltrating Lymphocytes. Clin Cancer Res; 24(17):4187-200
Gieras A, Gehbauer C, Perna-Barrull D, Engler JB, Diepenbruck I, Glau L, Joosse SA, Kersten N, Klinge S, Mittrücker HW, Friese MA, Vives-Pi M, Tolosa E. (2017) Prenatal administration of betamethasone causes changes in the T cell receptor repertoire influencing development of autoimmunity. Frontiers in Immunology. 8:1505.
Thom V, Schmid S, Gelderblom M, Hackbusch R, Kolster M, .., Pless O, Bernreuther C, Glatzel M, Wegscheider K, Gerloff C, Magnus T#, Tolosa E# (2016) IL-17 production by CSF lymphocytes as a biomarker for cerebral vasculitis. Neurol Neuroimmunol Neuroinflamm; 3(2):e214.
Rissiek A, Baumann I, Cuapio A, Mautner A, Kolster M, Arck PC, Dodge-Khatami A, Mittrücker HW, Koch-Nolte F, Haag F, Tolosa E (2015) The expression of CD39 on regulatory T cells is genetically driven and further upregulated at sites of inflammation. J Autoimmun; 58:12-20.
Adamopoulou E, Tenzer S, Hillen N, Klug P, Rota IA, Tietz S, Gebhardt M, Stevanovic S, Schild H, Tolosa E, Melms A, Stoeckle C (2013) Exploring the MHC-peptide matrix of central tolerance in the human thymus. Nat Commun. 4:2039. doi:10.1038/ncomms3039
Stoeckle C, Quecke P, Ruckrich T, Burster T, Reich M, Weber E, Kalbacher H, Driessen C, Melms A, Tolosa E (2012) Cathepsin S dominates autoantigen processing in human thymic dendritic cells. J Autoimmun. 38 (4):332-343.
Brucklacher-Waldert V, Stuerner K, Kolster M, Wolthausen J, Tolosa E (2009) Phenotypical and functional characterization of T helper 17 cells in multiple sclerosis. Brain. 132:3329-3341.
Feger U*, Tolosa E#, Huang Y-H, Waschbisch A, Biedermann T, Melms A, Wiendl H (2007) HLA-G expression defines a novel regulatory T-cell subset present in human peripheral blood and sites of inflammation. Blood. 110 (2):568-577.
Tolosa E, Li W, Yasuda Y, Wienhold W, Denzin LK, Lautwein A, Driessen C, Schnorrer P, Weber E, Stevanovic S, Kurek R, Melms A, Bromme D (2003) Cathepsin V is involved in the degradation of invariant chain in human thymus and is overexpressed in myasthenia gravis. Journal of Clinical Investigation. 112 (4):517-526.
Tolosa E, King LB, Ashwell JD (1998) Thymocyte Glucocorticoid Resistance Alters Positive Selection and Inhibits Autoimmunity and Lymphoproliferative Disease in MRL-lpr/lprMice. Immunity. 8 (1):67-76. 15.
#shared authorships
-
Vectored Immunotherapy (Dr. Anna Marei Mann)
Group members
Dr. Anna Marei Mann (née Eichhoff), Dr. Waldemar Schäfer, Nathalie Richter, Saruul Jargalsaikhan, Laura J. Becerra Moreno
Research interests
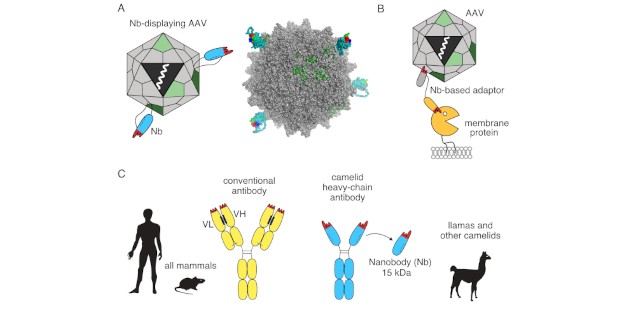
Adeno-associated viruses (AAV) are promising vectors for gene therapy due to their ability to infect a broad range of cells and tissues with minimal immunogenicity and toxicity but long-term expression of the transferred gene. A major challenge for therapeutic applications of AAV vectors is the low cell specificity of naturally occurring AAV serotypes and the high transduction of cells in the liver. Thus, high vector doses are often required to reach the tissue of interest, resulting in side effects and high production costs. We developed two novel technologies to enhance AAV-mediated transduction of target cells that express a specific membrane protein. One technology uses the genetic insertion of a membrane protein-specific nanobody (Nb) into the viral capsid (Fig. 1A), the other uses nanobody-based adaptor proteins (Fig. 1B). Nanobodies are the single variable immunoglobulin domains of heavy-chain antibodies from Camelids, i.e. alpacas and llamas. In contrast to conventional IgG antibodies, heavy chain antibodies do not associate with a light chain and their heavy chain lacks the CH1 domain (Fig. 1C). Nanobodies are used as targeting tools in research, diagnostics, imaging and therapy. The small size, high solubility and excellent stability account for the ease of reformatting.
Figure 1: The lab 'Vectored Immunotherapy' focuses on two strategies to retarget AAV vectors using membrane protein-specific nanobodies (Nbs). (A) Firstly, we retarget an AAV to a specific cell and/or tissue type by genetically inserting a membrane protein-specific nanobody into the viral capsid. The scheme depicts an AAV (grey/green) displaying a nanobody (Nb, cyan) on the capsid surface with a packaged DNA transgene (white) encoding a therapeutic gene product. The structural model of AAV9 (PDB ID: 3UX1) with a Nb inserted into a surface loop of VP1 was generated with AlphaFold and PyMol. (B) In a second strategy we retarget AAV to specific cells expressing the cognate membrane protein using a bispecific nanobody-based adaptor protein. One nanobody of the adaptor binds the AAV, the other nanobody binds a specific membrane protein. (C) Conventional antibodies (yellow) produced by all mammals are composed of two heavy and two light chains. The tip of two variable domains (red) (VH, VL) mediates specific binding to a target. In contrast, camelid heavy chain antibodies (cyan) consist only of heavy chains. Specific target binding is mediated by a single variable domain designated nanobody. Nanobodies are small (15 kDa), robust and highly soluble.
Current projects in the lab:
'nAAVi2mub - Nanobody-mediated AAV delivery to muscle and brain cells', research project funded by the Werner Otto Stiftung, PI: Dr. Anna Marei Mann.
Duchenne muscular dystrophy (DMD) is a fatal disease that manifests early during infancy or childhood and affects skeletal muscle, heart and diaphragm. This monogenic hereditary disorder is caused by a loss-of-function mutation. The recent approval of the AAV-based gene therapy 'Elevidys' for the treatment of DMD highlights the utility of AAV capsids with improved therapeutic efficacy and specificity. In this project, we insert nanobodies specific for an ecto-enzyme exclusively expressed on skeletal and heart muscle cells into receptor-blinded AAV2 and AAV9 variants. We evaluate the targeting specificity of such Nanobody-displaying AAV constructs in vitro on mouse and human muscle cell lines and in vivo in different mouse models.
'Nanobody-displaying AAV9 for the treatment of glioblastoma', part of the joint project THUNDER ('National Hub for Nanobody Cancer Theranostics') of the UKE and the UK Bonn funded by the German Cancer Aid (Deutsche Krebshilfe). Project-specific UKE partners: Prof. Friedrich Nolte (Inst. of Immunology); Prof. Peter Bannas, Dr. Julia Hambach (Radiology); Dr. Cecile Maire, Dr. Malte Mohme and Prof. Katrin Lamszus (Neurosurgery).
Glioblastoma is a devastating tumor characterized by a cold tumor microenvironment with inactive immune cells. In this project, we aim to use AAV vectors to express immune-activating cytokines (e.g. IL-12) and checkpoint- or purinergic signaling-inhibiting nanobodies (e.g. PD-L1 or CD73 and CD39) in order to promote an immune-activating tumor microenvironment. To efficiently transduce the tumor cells with AAV, we insert nanobodies specific for membrane proteins overexpressed by glioblastoma cells into the viral capsid of AAV9 and variants thereof.
'Nanobody-based adaptor proteins to retarget AAV vectors to specific cells' PI: Dr. Anna Marei Mann.
Our lab developed a technology to enhance AAV-mediated transduction of specific target cells using nanobody-based adaptor proteins. Such adaptors consist of two genetically fused nanobodies: an AAV-specific nanobody and a membrane protein-specific nanobody (Fig. 1B). We successfully selected nanobodies against various AAV serotypes including AAV1, 2, 4, 8 and 9 from immunized llama IgH-transgenic LamaMice (Eden et al. 2024, Nat. commun.). With these nanobodies we generated a variety of adaptors recognizing specific membrane proteins, including CD38, CD73, CD203a, CD296 and P2X7. We verified the capacity of these adaptors to enhance AAV-mediated transduction of cells expressing the cognate membrane protein. We aim to provide these adaptor proteins for academic and commercial partners as tools to improve the transduction of different cell types with various AAV serotypes.
'TransCAR: Transient CAR expression by targeted transfection of T lymphocytes with nanobody-targeted lipid nanoparticles (LNPs)', part of the transCAR consortium funded by the Landesforschungsförderung Hamburg, PIs: Dr. Julia Hambach, Dr. Anna Marei Mann.
A major goal of the transCAR consortium is to enable T cells and NK cells to express tumor-cell specific chimeric antigen receptors (CARs) using nanobody-targeted lipid nanoparticles (LNPs) that encapsulate a CAR-encoding mRNA. The goal of the project led by Dr. Julia Hambach and Dr. Anna Marei Mann is to generate nanobodies specific for membrane proteins expressed on T cells or NK cells, e.g. CD8 or CD16. Lipid nanoparticles will be conjugated with such nanobodies in order to specifically transfect T and NK cells.
Research networks
THUNDER ('National Hub for Nanobody Cancer Theranostics'), University Medical Center Hamburg-Eppendorf (UKE) and University Medical Center Bonn (UKB), funded by the German Cancer Aid.
Transiente CAR-Effektorzellen: Neue Ansätze zu ihrer Herstellung und effizienten Anwendung (TransCAR), UKE and UHH, funded by Landesforschungsförderung Hamburg.
Selected publications
Eden T, Schaffrath AZ, Wesolowski J, Stahler T, Tode N, Richter N, Schafer W, Hambach J, Hermans-Borgmeyer I, Woens J, Le Gall CM, Wendler S, Linke-Winnebeck C, Stobbe M, Budnicki I, Wanney A, Heitz Y, Schimmelpfennig L, Schweitzer L, Zimmer D, Stahl E, Seyfried F, Gebhardt AJ, Dieckow L, Riecken K, Fehse B, Bannas P, Magnus T, Verdoes M, Figdor CG, Hartlepp KF, Schleer H, Funer J, Tomas NM, Haag F, Rissiek B, Mann AM, Menzel S & Koch-Nolte F (2024). Generation of nanobodies from transgenic 'LamaMice' lacking an endogenous immunoglobulin repertoire. Nat Commun, 15: 4728.
Abad C, Demeules M, Guillou C, Gonde H, Zoubairi R, Tan YV, Pinto-Espinoza C, Schafer W, Mann AM, Vouret-Craviari V, Koch-Nolte F & Adriouch S (2024). Administration of an AAV vector coding for a P2X7-blocking nanobody-based biologic ameliorates colitis in mice. J Nanobiotechnology, 22: 27.
Hambach J, Mann AM, Bannas P & Koch-Nolte F (2022). Targeting multiple myeloma with nanobody-based heavy chain antibodies, bispecific killer cell engagers, chimeric antigen receptors, and nanobody-displaying AAV vectors. Front Immunol, 13: 1005800.
Mann AM, Schafer W, Adriouch S, Borner K, Grimm D, Braren I & Koch-Nolte F (2022). Enhanced Transduction of P2X7-Expressing Cells with Recombinant rAAV Vectors. Methods Mol Biol, 2510: 129-144.
Demeules M, Scarpitta A, Abad C, Gonde H, Hardet R, Pinto-Espinoza C, Eichhoff AM, Schafer W, Haag F, Koch-Nolte F & Adriouch S (2020). Evaluation of P2X7 Receptor Function in Tumor Contexts Using rAAV Vector and Nanobodies (AAVnano). Front Oncol, 10: 1699.
Eichhoff AM, Borner K, Albrecht B, Schafer W, Baum N, Haag F, Korbelin J, Trepel M, Braren I, Grimm D, Adriouch S & Koch-Nolte F (2019). Nanobody-Enhanced Targeting of AAV Gene Therapy Vectors. Mol Ther Methods Clin Dev, 15: 211-220.
Koch-Nolte F, Eichhoff A, Pinto-Espinoza C, Schwarz N, Schafer T, Menzel S, Haag F, Demeules M, Gonde H & Adriouch S (2019). Novel biologics targeting the P2X7 ion channel. Curr Opin Pharmacol, 47: 110-118.
Seifert L, Hoxha E, Eichhoff AM, Zahner G, Dehde S, Reinhard L, Koch-Nolte F, Stahl RaK & Tomas NM (2018). The Most N-Terminal Region of THSD7A Is the Predominant Target for Autoimmunity in THSD7A-Associated Membranous Nephropathy. J Am Soc Nephrol, 29: 1536-1548.
Menü