-
Calcium metabolism and cardiac contractility
Scientific activities
Dr. Felix Friedrich is a specialist in Clinical Pharmacology and Experimental Pharmacology and Toxicology. Since 2006 he teaches General and Special Pharmacology to students in medical studies, nursing and Study Nurses. He is the responsible of the Department of Experimental Pharmacology and Toxicology for the coordination of the Themenblock III and the Modul B in the human medical studies programs. In addition to research and teaching he is involved in pharmacologic counseling (Konsildienst). The scientific focus of the group is on the study of the genetic causes of hypertrophic cardiomyopathy (HCM), the resulting pathological mechanisms and new therapeutic approaches. In this context there is a close collaboration with the research group of Prof. Dr. Lucie Carrier. Hypertrophic cardiomyopathy is the most common congenital heart muscle disease (1: 500) and is caused in most cases by mutations in genes encoding for sarcomeric proteins. However, (40% ~) of patients may not have mutations in the already established HCM disease genes. Therefore, the identification of other disease-causing genes for a better understanding and treatment of HCM is highly relevant. HCM features are hypertrophy, diastolic dysfunction, loss of myocardial fibrosis and orientation. It is the main cause of sudden cardiac death in young adults and is associated with a significant risk of heart failure. Drug therapy remains largely empirical. In addition to the identification of additional disease genes a research focus is based on the evaluation of potential therapies for HCM. Our therapeutic approach is based on the hypothesis that a mutation-induced increased Ca2+ sensitivity of the myofilaments is a central pathomechanism of the disease. One aim of our projects is to evaluate the impact of substances that either directly affect the Ca2+ sensitivity of myofilaments or influence the intracellular Ca2+ homeostasis in HCM mouse models and heart-muscle samples of HCM patients. Target parameters are heart function assessed by echocardiography, cardiac morphology, molecular parameters and the Ca2+ sensitivity of ventricular muscle strips and contractile function of isolated myocytes. In this respect the group highly benefits from the expertise of the Department of Experimental and Clinical Pharmacology and Toxicology in the field of cardiovascular research.
Current projects
See also hypertrophic cardiomyopathy - AG Carrier
1. Screening for previously unknown gene mutations in patients with HCM and subsequent characterization of these mutants by viral overexpression in mouse cardiomyocytes and artificial heart tissue (engineered heart tissue, EHT) in cooperation with UKE HEXT AAV and O. Müller, Cardiology, Heidelberg.
2. Evaluation of potential therapies for HCM in the mouse model, on cardiac ventricular muscle strips, cardiomyocytes and the EHT model.
3. Evaluation of the role of proteins of the ubiquitin-proteasome system in HCM and other heart diseases.
4. Collaboration in the development of a clinical and experimental reference center of HCM in Northern Germany.
Application for thesis project
Prof. Dr. Eschenhagen, email: t.eschenhagen@uke.uni-hamburg.de
Current funding
Deutsche Stiftung für Herzforschung F-28-12, F-24-15
Publications
1. Friedrich FW, Bausero P, Sun Y, Treszl A, Krämer E, Juhr D, Richard P, Wegscheider K, Schwartz K, Brito D, Arbustini E, Waldenström A, Isnard R, Komajda M, Eschenhagen T, Carrier L; EUROGENE Heart Failure Project. A new polymorphism in human calmodulin III gene promoter is a potential modifier gene for familial hypertrophic cardiomyopathy. Eur Heart J. 2009 Jul;30(13):1648-55.
2. Friedrich FW, Wilding BR, Reischmann S, Crocini C, Lang P, Charron P, Müller OJ, McGrath MJ, Vollert I, Hansen A, Linke WA, Hengstenberg C, Bonne G, Morner S, Wichter T, Madeira H, Arbustini E, Eschenhagen T, Mitchell CA, Isnard R, Carrier L. Evidence for FHL1 as a novel disease gene for isolated hypertrophic cardiomyopathy. Hum Mol Genet. 2012 Jul 15;21(14):3237-54. doi: 10.1093/hmg/dds157.
3. Friedrich FW, Dilanian G, Khattar P, Juhr D, Gueneau L, Charron P, Fressart V, Vilquin JT, Isnard R, Gouya L, Richard P, Hammoudi N, Komajda M, Bonne G, Eschenhagen T, Dubourg O, Villard E, Carrier L. A novel genetic variant in the transcription factor Islet-1 exerts gain of function on myocyte enhancer factor 2C promoter activity. Eur J Heart Fail. 2013 Mar;15(3):267-76. doi: 10.1093/eurjhf/hfs178.
4. Friedrich FW, Reischmann S, Schwalm A, Unger A, Ramanujam D, Münch J, Müller OJ, Hengstenberg C, Galve E, Charron P, Linke WA, Engelhardt S, Patten M, Richard P, van der Velden J, Eschenhagen T, Isnard R, Carrier L. FHL2 expression and variants in hypertrophic cardiomyopathy. Basic Res Cardiol. 2014 Nov;109(6):451. doi: 10.1007/s00395-014-0451-8.
5. Flenner F*, Friedrich FW*, Ungeheuer N, Christ T, Geertz B, Reischmann S, Wagner S, Stathopoulou K, Söhren K, Weinberger F, Schwedhelm E, Cuello F, Maier LS, Eschenhagen T, Carrier L. Ranolazine improves tolerance to high workload by antagonizing β-adrenergic stimulation in cardiomyocytes of a mouse model of hypertrophic cardiomyopathy. Cardiovasc Res. 2015 Nov 3. pii: cvv247.
-
Cardiac hypertrophy/microRNAs
Team members
From left to right: Ann-Cathrin Kunze, Tessa Werner, Dr. Marc Hirt, Dr. Marita Rodriguez, Dr. Benjamin Kloth, Jutta Starbatty, Dr. Sandra Funcke
Addtional team members: Lena Bartholdt (also Pharmacoepigenetics), Jan-Tobias Hensel (also Pharmacoepigenetics), Aljosha Muttardi
Former members: Dr. Nils Sörensen, Dr. Jasper Boeddinghaus, Paul Demin
Scientific objectiv
Cardiac hypertrophy is the most important risk factor for the development of heart failure which is the leading cause of death in industrialized nations. We aim at deciphering some of the molecular mechanisms of cardiac hypertrophy by a model of cardiac hypertrophy which we have developed over the past couple of years. It is based on engineered heart tissue (EHT), a 3-dimensional cell culture format for heart cells.
Most fascinating is that engineered heart tissues (EHTs) beat spontaneously. In contrast to simple cell cultures of cardiomyocytes EHTs provide the opportunity to assess the essential cardiac properties frequency and force which can be analyzed automatically with technologies developed by the groups of Eschenhagen/Hansen in our institute.
Video-optical contractility analysis of an EHT
The idea behind our cardiac hypertrophy model is the enhancement of afterload/workload (AE = afterload enhancement) of these small tissue strips by a simple mechanical metal brace.
Video of a control EHT compared to an afterload enhanced EHT
And it works!
The model of cardiac hypertrophy displays in vitro many aspects of cardiac hypertrophy/heart failure: Cardiomyocyte hypertrophy, fibrosis, metabolic changes towards glycolysis, characteristic changes in coding and noncoding RNAs and many more.Literature:
Please find all technical details, our findings with the model, some other research we performed and cooperations in the publications below:
1. General practitioners' adherence to chronic heart failure guidelines regarding medication: the GP-HF study. Hirt MN, Muttardi A, Helms TM, van den Bussche H, Eschenhagen T. Clin Res Cardiol. 2015 Nov 9. [Epub ahead of print]; PMID:26552905
2. Deciphering the microRNA signature of pathological cardiac hypertrophy by engineered heart tissue- and sequencing-technology. Hirt MN, Werner T, Indenbirken D, Alawi M, Demin P, Kunze AC, Stenzig J, Starbatty J, Hansen A, Fiedler J, Thum T, Eschenhagen T. J Mol Cell Cardiol. 2015 Apr;81:1-9. doi: 10.1016/j.yjmcc.2015.01.008. [Epub ahead of print]; PMID: 25633833
3. Functional improvement and maturation of rat and human engineered heart tissue by chronic electrical stimulation. Hirt MN, Boeddinghaus J, Mitchell A, Schaaf S, Börnchen C, Müller C, Schulz H, Hubner N, Stenzig J, Stoehr A, Neuber C, Eder A, Luther PK, Hansen A, Eschenhagen T. J Mol Cell Cardiol. 2014 Sep;74:151-61. doi: 10.1016/j.yjmcc.2014.05.009. Epub 2014 May 19.; PMID: 24852842
4. Automated analysis of contractile force and Ca2+ transients in engineered heart tissue. Stoehr A, Neuber C, Baldauf C, Vollert I, Friedrich FW, Flenner F, Carrier L, Eder A, Schaaf S, Hirt MN, Aksehirlioglu B, Tong CW, Moretti A, Eschenhagen T, Hansen A. Am J Physiol Heart Circ Physiol. 2014 May;306(9):H1353-63. doi: 10.1152/ajpheart.00705.2013. Epub 2014 Feb 28.; PMID: 24585781
5. Cardiac tissue engineering: state of the art. Hirt MN, Hansen A, Eschenhagen T. Circ Res. 2014 Jan 17;114(2):354-67. doi: 10.1161/CIRCRESAHA.114.300522. Review. PMID: 24436431
6. Contractile abnormalities and altered drug response in engineered heart tissue from Mybpc3-targeted knock-in mice. Stöhr A, Friedrich FW, Flenner F, Geertz B, Eder A, Schaaf S, Hirt MN, Uebeler J, Schlossarek S, Carrier L, Hansen A, Eschenhagen T. J Mol Cell Cardiol. 2013 Oct;63:189-98. doi: 10.1016/j.yjmcc.2013.07.011. Epub 2013 Jul 26.; PMID: 23896226
7. Increased afterload induces pathological cardiac hypertrophy: a new in vitro model. Hirt MN, Sörensen NA, Bartholdt LM, Boeddinghaus J, Schaaf S, Eder A, Vollert I, Stöhr A, Schulze T, Witten A, Stoll M, Hansen A, Eschenhagen T. Basic Res Cardiol. 2012 Nov;107(6):307. doi: 10.1007/s00395-012-0307-z. Epub 2012 Oct 26.; PMID: 23099820
8. Human engineered heart tissue as a versatile tool in basic research and preclinical toxicology. Schaaf S, Shibamiya A, Mewe M, Eder A, Stöhr A, Hirt MN, Rau T, Zimmermann WH, Conradi L, Eschenhagen T, Hansen A. PLoS One. 2011;6(10):e26397. doi: 10.1371/journal.pone.0026397. Epub 2011 Oct 20.; PMID: 22028871
9. Vernakalant: a novel antiarrhythmic drug for the rapid conversion of atrial fibrillation to sinus rhythm. Hirt MN, Eschenhagen T. Dtsch Med Wochenschr. 2010 May;135(19):971-6. doi: 10.1055/s-0030-1253686. Epub 2010 May 5. Review. German.; PMID: 20446233
10. Kidney injury molecule 1 (Kim1) is a novel ciliary molecule and interactor of polycystin 2. Kuehn EW, Hirt MN, John AK, Muehlenhardt P, Boehlke C, Pütz M, Kramer-Zucker AG, Bashkurov M, van de Weyer PS, Kotsis F, Walz G. Biochem Biophys Res Commun. 2007 Dec 28;364(4):861-6. doi: 10.1016/j.bbrc.2007.10.103. Epub 2007 Oct 26.; PMID: 18273441
-
Cardiac cellular electrophysiology
We are interested in electrophysiological mechanisms involved in electrical refractoriness and generation of contractile force in human heart muscle. Therefore, we measure ionic currents in isolated cells and action potentials and force in intact heart muscle preparations. One focus is on chronic atrial fibrillation (AF). We address the question how AF modifies the electrophysiological properties of human atrium. We try to find out reasons for the distorted excitation-contraction coupling in AF. In order to identify new targets for anti-AF drugs, we measure effects of blocking of ion channels that could serve potentially as targets in AF. Finally, we study effects by adrenergic activation, on cardiac tissues from humans and other species. Another focus is on human induced pluripotent stem cell derived cardiomyocytes (hIPSC). Availability of hIPSC-CM may offer astonishing options. They could be used therapeutically and/or as a pharmacological model. Since hPISC-CM are of human origin, they should represent human-specific electrophysiological properties. However, despite the fact that all major ion currents of human adult heart are expressed in hIPSC exact knowledge about regulation of refractoriness and contractility in hIPSC is spare. Therefore, we compare electrophysiological properties of hIPSC-CM to those of mature human cardiomyocytes.
-
Hypertrophic cardiomyopathy
Genetics, Pathophysiology and Therapy of Hypertrophic Cardiomyopathy
Team leader
Prof. Dr. Lucie Carrier 57208 l.carrier@uke.de
Researchers
Dr. Giulia Mearini 53180 g.mearini@uke.de
Dr. Felix Friedrich 53180 f.friedrich@uke.de
Dr. Saskia Schlossarek 54707 s.schlossarek@uke.de
Dr. Frederik Flenner 58597 f.flenner@uke.de
Dr. Verena Behrens-Gawlik 52179 v.behrens@uke.de
Technicians
Elisabeth Krämer 58597 e.kraemer@uke.de
Silke Reischmann 52179 s.reischmann@uke.deBirgit Geertz 54707 b.geertz@uke.de
PhD Students
Sonia Singh 58597 s.singh@uke.de
Maksymilian Prondzynski 58597 m.prondzynski@uke.de
Antonia Zech 59770 a.zech@uke.de
MD students
Nico Kresin 57291 Nico.Kresin@stud.uke.uni-hamburg.de
Marc Philip von See 59770 Marc-Philip.von-See@stud.uke.uni-hamburg.de
-
Engineered heart tissue
Tissue engineering and stem cells - Engineered heart tissue
The techniques to functionally characterize cardiomyocytes in vitro are limited. The focus of the EHT team is the establishment and optimization of techniques to engineer three-dimensional, force-generating heart tissue from single cells and develop test platforms and protocols to analyze relevant parameters of cardiomyocyte biology. The work is based on our long term engagement in cardiac tissue engineering. We previously established protocols to generate EHTs from neonatal mouse/rat heart cells or cardiomyocytes from differentiated human pluripotent stem cells and test systems to analyze contractility and calcium transients with a high level of automation and reproducibility. The EHT technology is specifically used in predictive toxicology, disease modeling and cardiac repair.
Predictive toxicology
The second most frequent reason for late stage drug failure is cardiac toxicity. The analysis of cardiac toxicity during preclinical drug development is compromised in part due to unavailability of non-failing human heart tissue. We have shown in proof-of-principle experiments in neonatal rat cardiac myocytes (NRCM) that inhibitors of repolarisation lead to a concentration-dependent prolongation of EHT relaxation. Whereas these effects in rat EHTs are independent of hERG, selective hERG channel inhibitors exert relaxation-prolongation and arrhythmias in human EHTs precisely matching findings in hERG channel assays. We are evaluating if the EHT technology is a meaningful addition to the toolset of in vitro assays. In addition, we study the question if the parallel analysis of contractility, voltage and calcium transients (by genetically encoded sensors) is increasing the predictive value of this technology.
Disease modeling
Understanding the pathophysiology of cardiac diseases is incomplete and drug development programs are patchy. Our aim is to establish high content-in vitro models for important acquired and inherited cardiac diseases. In this context we are optimizing protocols to reprogram hiPSCs from fibroblasts from patients with hereditary cardiac diseases, to generate isogenic controls (by CRISPR/Cas9 genomic editing), to generate cardiomyocytes, studythe disease-specific phenotype in EHTs and try new therapeutic options in vitro. We described the phenotype of two mouse models of hypertrophic cardiomyopathy (cMybpC-knockout/knockin) in neonatal mouse cardiomyocyte-EHTs. In parallel we established a small library of human iPS cells from patients with hereditary cardiac diseases (Long QT syndrome, Brugada syndrome, catecholaminergic polymorphic ventricular tachycardia, hypertrophic cardiomyopathy, dilated cardiomyopathy) and study the disease-specific phenotype in EHTs. Furthermore we have optimized protocols to induce pathological cardiac hypertrophy by afterload enhancement - the most frequent cause of heart failure - in rat EHT together with the group cardiac hypertrophy. We are furthermore transferring this technology to EHT from human iPS cell-derived cardiomyocytes with projects like the ERC-funded project IndivuHeart. Moreover, epigenetic modifier drugs are evaluated in the same system in collaboration with the group Pharmacoepigenetics.
Methods
- Generation of EHT in 24 and 6 well-format from neonatal mouse and rat heart cells and cardiomyocytes from human pluripotent stem cells
- Differentiation of human pluripotent stem cells into cardiomyocytes
- Automated analysis of contractility and calcium transients under baseline conditions and pharmacological intervention
- Genome editing by CRISPR/Cas9
- Transduction of cardiomyocytes with Adeno-associated virus (AAV) and lentivirus
- Transplantation of EHT onto the left ventricle of small animals
Funding
- Deutsches Zentrum für Herz-Kreislauf-Forschung e.V.
- Deutsche Forschungsgemeinschaft
- Europäische Union
- Deutsche Stiftung für Herzforschung
- Freie und Hansestadt [IM1] Hamburg, Behörde für Wissenschaft und Forschung
Selected Publications
- Eschenhagen T, Fink C, Remmers U, Scholz H, Wattchow J, Weil J, Zimmermann W, Dohmen HH, Schäfer H, Bishopric N, Wakatsuki T, Elson EL. Three-dimensional reconstitution of embryonic cardiomyocytes in a collagen matrix: a new heart muscle model system. The FASEB Journal: official publication of the Federation of American Societies for Experimental Biology. 1997; 11:683-94.
- Zimmermann WH, Fink C, Kralisch D, Remmers U, Weil J, Eschenhagen T. Three-dimensional engineered heart tissue from neonatal rat cardiac myocytes. Biotechnology and bioengineering. 2000; 68:106-14.
- Zimmermann W-H, Schneiderbanger K, Schubert P, Didié M, Münzel F, Heubach JF, Kostin S, Neuhuber WL, Eschenhagen T. Tissue engineering of a differentiated cardiac muscle construct. Circulation Research. 2002; 90:223-30.
- Hansen A, Eder A, Bönstrup M, Flato M, Mewe M, Schaaf S, Aksehirlioglu B, Schwörer A, Uebeler J, Eschenhagen T. Development of a Drug Screening Platform Based on Engineered Heart Tissue. Circulation Research. 2010;
- Schaaf S, Shibamiya A, Mewe M, Eder A, Stöhr A, Hirt MN, Rau T, Zimmermann W-H, Conradi L, Eschenhagen T, Hansen A. Human engineered heart tissue as a versatile tool in basic research and preclinical toxicology. PloS one. 2011; 6:e26397.
- Hirt MN, Sörensen NA, Bartholdt LM, Boeddinghaus J, Schaaf S, Eder A, Vollert I, Stöhr A, Schulze T, Witten A, Stoll M, Hansen A, Eschenhagen T. Increased afterload induces pathological cardiac hypertrophy: a new in vitro model. Basic research in cardiology. 2012; 107:307.
- Zimmermann W-H, Melnychenko I, Wasmeier G, Didié M, Naito H, Nixdorff U, Hess A, Budinsky L, Brune K, Michaelis B, Dhein S, Schwoerer A, Ehmke H, Eschenhagen T. Engineered heart tissue grafts improve systolic and diastolic function in infarcted rat hearts. Nature Medicine. 2006; 12:452-8.
-
Pharmacoepigenetics
The group focuses on pharmacoepigenetics and closely collaborates with Marc Hirt ( cardiac hypertrophy/micro RNAs ) and Arne Hansen ( Engineered Heart Tissue ) and their groups from the same department as well as the group of Tanja Zeller and Julia Krause at the University Heart Center Hamburg.
Epigenetics evolved as a generic term to describe all chromatin modifications that have medium- or long-term impact on transcriptional regulation. Among others these modifications comprise histone and DNA modifications such as histone acetylation, methylation, phosphorylation or sumoylation or DNA methylation and hydroxymethylation respectively. On the one hand these regulatory marks can be stable over years and even inherited. On the other hand they can also be highly dynamic and be influenced by drugs.
Transcriptional dysregulation is an important feature of cardiac hypertrophy and heart failure. Understanding the role of epigenetics for this transcriptional regulation and dysregulation in hypertrophy and how it can be pharmacologically influenced might improve the success of heart failure of atrial fibrillation drug therapy, with the potential of treating the cause of the disease rather than the symptoms.
Our research is currently centered on DNA methylation. We are investigating different components of the cellular methylation and demethylation machinery in the context of the department's well established in vitro tissue culture model EHT (engineered heart tissue). This model is based on both atrial and ventricular cardiomyocytes derived from human induced pluripotent stem cells (hiPS-cells). Using this model, we are working on the function of DNA methyltransferase 3 a (DNMT3A) in pathological hypertrophy, the role of epigenetics in atrial fibrillation and fibrosis and we are trying to harness DNA methylation therapeutically in an epigenome editing approach.
Our fibroblast project is embedded in a collaboration with researchers in Milano, Montpellier and Madrid. We are moreover collaborating with the group of Roger Foo at the Genome Institute of Singapore.The group is additionally funded by the German Research Foundation (DFG) and the european initiative ERA-CVD (European Research Area Network Cardiovascular Disaese).
-
Molecular pharmacology
AG Molecular Pharmacology
From left to right: Mara Goetz, Dr. Sonia Donzelli, Prof. Dr. Friederike Cuello, Angelika Piasecki, cand. rer.nat. Simon Diering, Dr. Konstantina Stathopoulou, Dr. Sophie Schobesberger, Steven Schulz MSc.
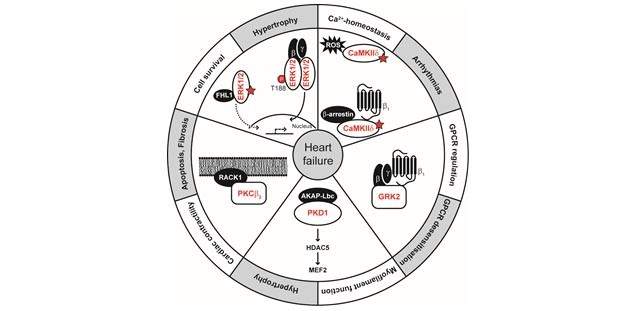
Scientific Interest: The group investigates the basic molecular mechanisms leading to alterations in signal transduction pathways during cardiovascular disease development. The focus of the research projects continues to be the analysis of disease-specific post-translational modification patterns. This involves for example dephosphorylation, oxidative modifications such as S-glutathiolation and inter-and interdisulfide formation in redox sensor proteins and the resulting consequences for myofilament function and protein kinase activity. Among the various standard biochemical, molecular biological and physiological techniques applied is for example the isolation, culture and viral infection of adult and neonatal cardiac myocytes, DNA-manipulation, histology, immunocytochemistry, confocal microscopy, protein kinase activity assays by FRET, second messenger binding assays and redox proteomics analysis.
Selected Publications:
2015
- Flenner F, Friedrich FW, Ungeheuer N, Christ T, Geertz B, Reischmann S, Wagner S, Stathopoulou K, Sohren KD, Weinberger F, Schwedhelm E, Cuello F, Maier LS, Eschenhagen T, Carrier L. Ranolazine antagonizes catecholamine-induced dysfunction in isolated cardiomyocytes, but lacks long-term therapeutic effects in vivo in a mouse model of hypertrophic cardiomyopathy. Cardiovasc Res. 2015
- Carrier L, Mearini G, Stathopoulou K, Cuello F. Cardiac myosin-binding protein c (MYBPC3) in cardiac pathophysiology. Gene. 2015;573:188-197 2014 Lorenz K, Stathopoulou K, Schmid E, Eder P, Cuello F. Heart failure-specific changes in protein kinase signalling. Pflugers Arch. 2014;466:1151-1162
- Cuello F, Shankar-Hari M, Mayr U, Yin X, Marshall M, Suna G, Willeit P, Langley SR, Jayawardhana T, Zeller T, Terblanche M, Shah AM, Mayr M. Redox state of pentraxin 3 as a novel biomarker for resolution of inflammation and survival in sepsis. Mol Cell Proteomics. 2014;13:2545-2557
- Candasamy AJ, Haworth RS, Cuello F, Ibrahim M, Aravamudhan S, Kruger M, Holt MR, Terracciano CM, Mayr M, Gautel M, Avkiran M. Phosphoregulation of the titin-cap protein telethonin in cardiac myocytes. J Biol Chem. 2014;289:1282-1293
- Stathopoulou K, Cuello F, Candasamy AJ, Kemp EM, Ehler E, Haworth RS, Avkiran M. Four-and-a-half LIM domains proteins are novel regulators of the protein kinase D pathway in cardiac myocytes. Biochem J. 2014;457:451-461
- Holland RJ, Paulisch R, Cao Z, Keefer LK, Saavedra JE, Donzelli S. Enzymatic generation of the NO/HNO-releasing IPA/NO anion at controlled rates in physiological media using β-galactosidase. Nitric Oxide. 2013;35:131-136
- Donzelli S, Fischer G, King BS, Niemann C, DuMond JF, Heeren J, Wieboldt H, Baldus S, Gerloff C, Eschenhagen T, Carrier L, Boger RH, Espey MG. Pharmacological characterization of 1-nitrosocyclohexyl acetate, a long-acting nitroxyl donor that shows vasorelaxant and antiaggregatory effects. J Pharmacol Exp Ther. 2013;344:339-347
- Abonnenc M, Nabeebaccus AA, Mayr U, Barallobre-Barreiro J, Dong X, Cuello F, Sur S, Drozdov I, Langley SR, Lu R, Stathopoulou K, Didangelos A, Yin X, Zimmermann WH, Shah AM, Zampetaki A, Mayr M. Extracellular matrix secretion by cardiac fibroblasts: Role of microRNA-29b and microRNA-30c. Circ Res. 2013;113:1138-1147
- Fraysse B, Weinberger F, Bardswell SC, Cuello F, Vignier N, Geertz B, Starbatty J, Kramer E, Coirault C, Eschenhagen T, Kentish JC, Avkiran M, Carrier L. Increased myofilament Ca2+ sensitivity and diastolic dysfunction as early consequences of Mybpc3 mutation in heterozygous knock-in mice. J Mol Cell Cardiol. 2012;52:1299-1307
- Bardswell SC, Cuello F, Kentish JC, Avkiran M. cMyBP-C as a promiscuous substrate: phosphorylation by non-PKA kinases and its potential significance. J Muscle Res Cell Motil. 2012;33:53-60
- Sadayappan S, Gulick J, Osinska H, Barefield D, Cuello F, Avkiran M, Lasko VM, Lorenz JN, Maillet M, Martin JL, Brown JH, Bers DM, Molkentin JD, James J, Robbins J. A critical function for Ser-282 in cardiac myosin binding protein-c phosphorylation and cardiac function. Circ Res. 2011;109:141-150
- Makowski MR, Wiethoff AJ, Blume U, Cuello F, Warley A, Jansen CH, Nagel E, Razavi R, Onthank DC, Cesati RR, Marber MS, Schaeffter T, Smith A, Robinson SP, Botnar RM. Assessment of atherosclerotic plaque burden with an elastin-specific magnetic resonance contrast agent. Nat Med. 2011;17:383-388
- Haworth RS, Cuello F, Avkiran M. Regulation by phosphodiesterase isoforms of protein kinase A-mediated attenuation of myocardial protein kinase D activation. Basic Res Cardiol. 2011;106:51-63 Grube K, Rudebusch J, Xu Z, Bockenholt T, Methner C, Muller T, Cuello F, Zimmermann K, Yang X, Felix SB, Cohen MV, Downey JM, Krieg T. Evidence for an intracellular localization of the adenosine A2B receptor in rat cardiomyocytes. Basic Res Cardiol. 2011;106:385-396
- Cuello F, Bardswell SC, Haworth RS, Ehler E, Sadayappan S, Kentish JC, Avkiran M. Novel role for p90 ribosomal s6 kinase in the regulation of cardiac myofilament phosphorylation. J Biol Chem. 2011;286:5300-5310
- Yin X, Cuello F, Mayr U, Hao Z, Hornshaw M, Ehler E, Avkiran M, Mayr M. Proteomics analysis of the cardiac myofilament subproteome reveals dynamic alterations in phosphatase subunit distribution. Mol Cell Proteomics. 2010;9:497-509
- Mattock KL, Gough PJ, Humphries J, Burnand K, Patel L, Suckling KE, Cuello F, Watts C, Gautel M, Avkiran M, Smith A. Legumain and cathepsin-l expression in human unstable carotid plaque. Atherosclerosis. 2010;208:83-89
- Madhani M, Hall AR, Cuello F, Charles RL, Burgoyne JR, Fuller W, Hobbs AJ, Shattock MJ, Eaton P. Phospholemman Ser69 phosphorylation contributes to sildenafil-induced cardioprotection against reperfusion injury. Am J Physiol Heart Circ Physiol. 2010;299:H827-836
- El-Armouche A, Wahab A, Wittkopper K, Schulze T, Bottcher F, Pohlmann L, King SB, DuMond JF, Gerloff C, Boger RH, Eschenhagen T, Carrier L, Donzelli S. The new HNO donor, 1-nitrosocyclohexyl acetate, increases contractile force in normal and β-adrenergically desensitized ventricular myocytes. Biochem Biophys Res Commun. 2010;402:340-344
- Bardswell SC, Cuello F, Rowland AJ, Sadayappan S, Robbins J, Gautel M, Walker JW, Kentish JC, Avkiran M. Distinct sarcomeric substrates are responsible for protein kinase D-mediated regulation of cardiac myofilament Ca2+ sensitivity and cross-bridge cycling. J Biol Chem. 2010;285:5674-5682
-
Cardiac Repair
Cardiac Regeneration and Repair
Myocardial damage following myocardial infarction or other cardiac diseases results in loss of working myocardium and subsequent heart failure. Despite new therapies prognosis for heart failure remains poor, and new therapeutic options are urgently needed. In this context, regenerative approaches have gained increasing attention.
Cardiac regeneration
Adult cardiac myocytes were classically regarded as withdrawn from the cell cycle. This dogma has been challenged by recent findings suggesting that a very small fraction of adult cardiomyocytes in human heart is regenerated. At present it is unclear whether this regeneration occurs from an undifferentiated precursor cell population or from cardiomyocytes. The existence of a cardiac precursor pool within the adult heart remains ambiguous, but obviously such a cardiac precursor cells would be particularly attractive for therapeutic strategies. Different cardiac precursor cell types have been characterized by the expression of stem or progenitor cell marker proteins.
Cardiac repair
A different therapeutical appraoch is the in vitro generation of cardiac tissue (engineered heart tissue), which in a second step, can be used for cardiac repair. The major limitation for this strategy so far, was the lack of human cardiomyocytes. The possibility of generating unlimited numbers of human cardiomyocytes from human embryonic as well as human induced pluripotent stem cells has opened the door for future clinical application of these constructs. We are currently working on different projects to further evaluate the possibility to use these constructs for tissue replacement therapy in different disease models.
Methods
- Induced pluripotent stem cell culture (iPSC culture)
- 2D and 3D cardiac differentiation of iPSCs
- Functional EHT measurements
- Fluorescence microspcopy
- in vivo models of cardiac ischemia
- CRISPR/Cas9
Recent Publications
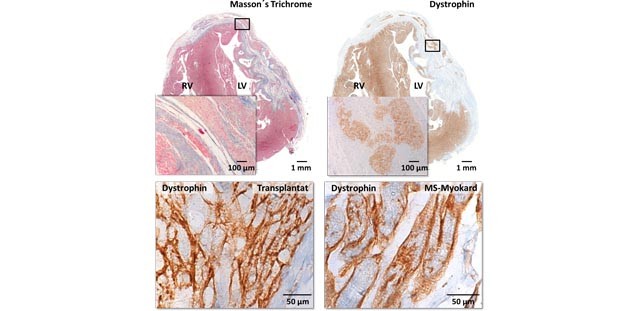
Implantation of hiPSC-derived Cardiac-muscle Patches after Myocardial Injury in a Guinea Pig Model. Castro L, Geertz B, Reinsch M, Aksehirlioglu B, Hansen A, Eschenhagen T, Reichenspurner H, Weinberger F, Pecha S. J Vis Exp. 2019 Mar 18;(145). doi: 10.3791/58810
Evidence for Minimal Cardiogenic Potential of Stem Cell Antigen 1-Positive Cells in the Adult Mouse Heart. Neidig LE, Weinberger F, Palpant NJ, Mignone J, Martinson AM, Sorensen DW, Bender I, Nemoto N, Reinecke H, Pabon L, Molkentin JD, Murry CE, van Berlo JH. Circulation. 2018 Dec 18;138(25):2960-2962. doi: 10.1161/CIRCULATIONAHA.118.035273. doi: 10.1161/CIRCULATIONAHA.118.035273
No effect of thymosin beta-4 on the expression of the transcription factor Islet-1 in the adult murine heart. Weinberger F, Nicol P, Starbatty J, Stubbendorff M, Becher PM, Schrepfer S, Eschenhagen T. Pharmacol Res Perspect. 2018 Jun;6(3):e00407. doi: 10.1002/prp2.407.
Diltiazem prevents stress-induced contractile deficits in cardiomyocytes, but does not reverse the cardiomyopathy phenotype in Mybpc3-knock-in mice. Flenner F, Geertz B, Reischmann-Düsener S, Weinberger F, Eschenhagen T, Carrier L, Friedrich FW. J Physiol. 2017 Jun 15;595(12):3987-3999. doi: 10.1113/JP273769.
Cardiac repair in guinea pigs with human engineered heart tissue from induced pluripotent stem cells. Weinberger F, Breckwoldt K, Pecha S, Kelly A, Geertz B, Starbatty J, Yorgan T, Cheng KH, Lessmann K, Stolen T, Scherrer-Crosbie M, Smith G, Reichenspurner H, Hansen A, Eschenhagen T. Sci Transl Med. 2016 Nov 2;8(363):363ra148.
Menü