LARGE PUBLIC-PRIVATE CONSORTIUM
The ELBS is a dynamic network consisting of partners from academia and industry with the common goal of bringing LB tests into clinical routine. The network covers all relevant areas to reach this goal, ranging from harmonization of technologies, to the design of suitable clinical studies, and the interaction with regulatory bodies. The combination of the collective expertize and knowledge of the ELBS members with the strategic implementation of joint projects makes the ELBS a uniquely suited driver and valuable partner in the area of LB.
CONSORTIUM SIZE AND DIVERSITY
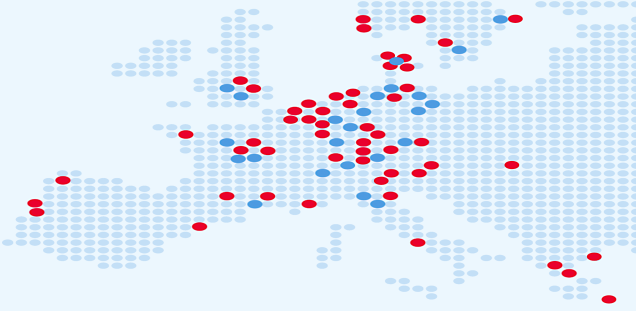
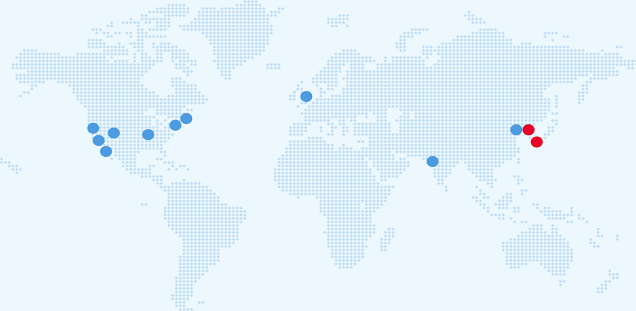
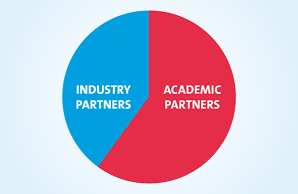
The ELBS network currently consists of more than 100 member institutions with more joining each year. About 60 percent of our members are academic institutions or university hospitals; About 40 percent represent industry partners spread across biotech, device manufacturers, diagnostic companies, standard providers and pharmaceutical companies. The ELBS member institutions also span four continents (Asia, Europe, USA, Australia), the majority of members being located in Europe.
HARMONIZATION AND STANDARDIZATION
In order to work towards robust LB tests that are applicable in a clinical setting, standardization and harmonization of preanalytical as well as analytical parameters are essential. One valuable approach to achieve such harmonization is the formulation of specific guidelines. Another is performing actual wet lab work including ring experiments and technology benchmarking. The ELBS includes both complementary approaches in its activity profile.
COMPLEMENTARY FOCUS AREAS BY DIFFERENT WORKING GROUPS
In order to move the field of LB forward and in line with our holistic approach, the ELBS consists of distinct working groups that combine specific areas of expertize. The working groups (WGs) are Dissemination & Education, Clinical, Technology, and Regulatory Issues. Each WG is chaired by a steering committee of 2 – 4 members that drive the activity of the network in their specific area of expertize. More information on the WGs can be found under the respective links below.