Research Focus
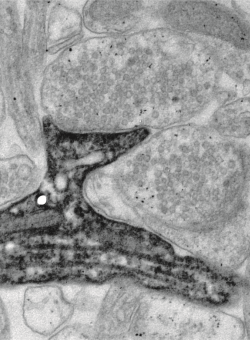
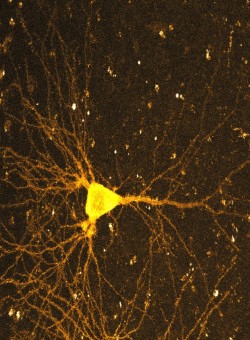
Neurons in the brain communicate through chemical synapses. Depending on the activity of pre- and postsynaptic cells, these communication channels can rapidly and persistently change their strength. These functional adaptations, collectively known as long-term plasticity, involve a large number of intracellular signaling systems. On longer timescales, new synapses are established between previously unconnected cells while other synaptic connections are completely removed. Together, these changes in the efficacy and connectivity of brain circuits are thought to be crucial for information processing and memory storage in the brain.
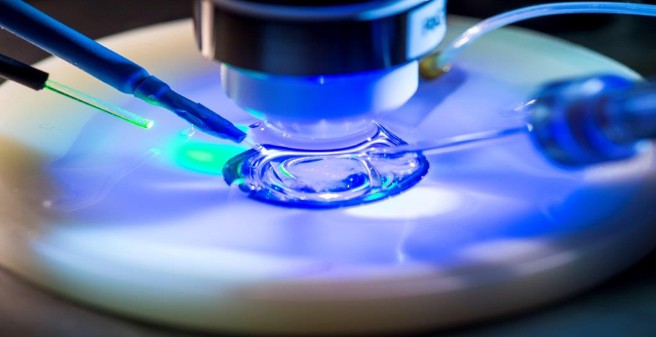
We develop optogenetic methods to stimulate identified neurons and to optically measure the amplitude of postsynaptic calcium transients in dendritic spines. Two-photon laser scanning microscopy allows us to perform such optophysiological experiments in intact brain tissue with high spatial and temporal resolution. Using genetically encoded probes, we monitor the activity of single synapses over several hundred stimulations and measure parameters such as synaptic potency and the probability of glutamate release. Optical induction of plasticity at individual, identified synapses allows us to investigate the underlying electrical and biochemical processes in great detail. The connectivity of our brain constantly changes in response to sensory experience (Huber et al., 2012). A central aim of our research is to understand the rules and molecular mechanisms that govern our extraordinary ability to learn and to remember.
Current Projects
Second messenger systems regulating synaptic plasticity
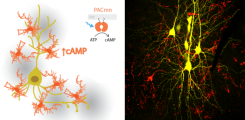
I would like to understand how networks of neurons and their synaptic connections store information. Towards reaching this goal, we have developed an all-optical method to induce spike-timing-dependent plasticity at a small subset of hippocampal CA3-CA1 connections so that we can follow the changes in synaptic strength over days (Anisimova et al., 2021). I also am very interested in modulation of synaptic strength by G-protein-coupled receptors and intracellular signaling cascades downstream of cAMP and cGMP. To bypass the receptors and study the intracellular cascades in particular cells, I have been involved in developing and characterizing new optogenetic tools such as the rhodopsin guanylyl and adenylyl cyclases (Scheib et al., 2015, Scheib et al., 2018), and a membrane-anchored no dark activity photoactivatable adenylyl cyclase called PACmn (Constantin et al., 2021). SynTagMA (Perez-Alvarez et al., 2020) and a light-activated potassium channel (Beck et al., 2018) are other tools that have become quite useful for studying and modulating network activity. Using some of these optogenetic tools has revealed a key role of unexpected inter-cellular communication pathways downstream of cAMP or neuronal spiking that we are working hard to understand. My work is currently funded by the Landesforschungsförderung Hamburg and the DFG (FOR 2419 and SFB 1328).
Christine Gee
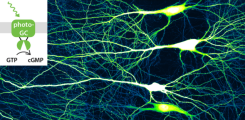
Role of cyclic nucleotides in synaptic function
The goal of my project is to elucidate the role of cyclic nucleotides (cAMP and cGMP) in synaptic function. Alterations in synaptic strength are thought to be the cellular correlate of learning. These alterations can occur during long-term potentiation, in which the roles of cAMP and cGMP are still unclear. Especially in the hippocampus, not much is known of the cell specific role of cGMP in synaptic function. I am using genetically encoded sensors and photoactivatable cyclases such as BeCyclOp (Gao et al. 2015) as well as patch-clamp recordings to assess cyclic nucleotide dynamics and their impact on synaptic strength in different cell types.
Marie Eileen Wiesenhavern
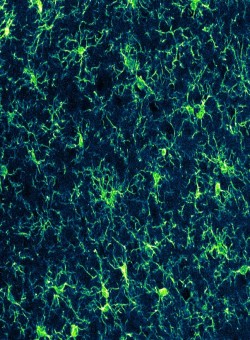
Second messenger signaling in microglia
While the role of microglial second messenger signaling in learning and memory is established, the focus has been on the effects of intracellular calcium. In my project, I am expanding the toolbox for optogenetic manipulations of microglia by the development of a microglia-specific PACmn mouse line. Using blue light as stimulation, intracellular cAMP can be elevated specifically in microglia with high spatio-temporal resolution. This development enables the investigation of microglial cyclic nucleotide signaling and its effects on microglial properties, as well as on microglia-neuron interaction.
Oana Constantin
Development of optogenetic enzymes
Cyclic AMP is one of the most important intracellular signaling molecules, its increase and decrease governs a large repertoire of cellular functions. It is an ancient molecule that can be found in organisms from all kingdoms of life, from bacteria to plants and animals. We develop tools to modulate the intracellular concentration of cAMP with high spatial and temporal resolution, which is not possible with traditional pharmacological approaches. Improved light-activated adenylyl cyclases (CaRhAC, PACmn) can be selectively expressed in the cells of interest and activated with light to tightly control intracellular cAMP levels. In the brain, cyclic AMP is thought to be involved in learning: When changes occur in the environment, the connections between specific neurons strengthen, a process known as long-term plasticity or LTP. Cyclic AMP signaling pathways are active during the consolidation of LTP, a process that is dependent on protein synthesis and structural changes at synapses. I am using the newly developed optogenetic tools to unravel the function of cAMP signaling in the hippocampus during long term plasticity.
Oana Constantin & Carolina Pinto
Interactions of microglia and neurons after stroke
Stroke is one of the highest causes of death and disability world-wide. Despite many years searching for neuroprotective therapies, re-establishing perfusion to the affected area remains the only effective treatment.
Microglia, commonly referred to as the immune cell of the central nervous system, are involved in post-stroke processes. They migrate into the affected area where they clear debris and drive a strong neuroinflammatory response. We are interested in if manipulating microglial function can affect recovery after stroke.
We express a Gq-DREADD (Designer Receptor Exclusively Activated by Designer Drugs) only in microglia using the CreERT2 system, and stimulate Gq signaling with CNO and investigate the effect on stroke outcome.
Charlotte Oldenburg
The role of microglia in the recovery after stroke
In the recovery period after a stroke, lost motor function is slowly regained over a period of several weeks. This process involves changes at the synaptic level, but it not known whether microglia activity helps or hinders this process. In genetically modified mice, we control G-protein signaling pathways in microglia with chemogenetic tools to asses the impact on the recovery process. This project, a collaboration with Tim Magnus’ group at the UKE, is supported by the DFG (SPP 2395).
Marie Luise Brehme & Charlotte Oldenburg
Neighboring synapses: Collaboration or competition?
The formation and elimination of dendritic spines play an important role in structural remodeling of neuronal networks. This process is highly dynamic and occurs over different timescales. To get a better understanding of the dynamics of spine formation and interaction between nearby spines, we image excitatory and inhibitory synapses over extended time periods up to several days using organotypic brain slice cultures as a model. We are particularly interested in the removal of spines and the appearance of novel spines: Do these events have any repercussions in the local dendritic neighborhood? Are they shaped by homeostatic mechanisms?
Kristina Ponimaskine
Changes in synaptic ultrastructure after LTP
Synaptic plasticity events, dynamic changes in the efficacy of synapses, represent a major mechanism of information storage in the brain. Importantly, synaptic plasticity is not only electrochemical but also structural and mechanical in nature, an aspect that is often called ‘structural’ plasticity to distinguish it from functional read-out. In my project, I’m particularly interested in studying the ultrastructural plasticity to reveal the material basis of information storage at synapses. I generate the information by manipulating the synapse-of-interest optogenetically, followed by electron microscope (EM) imaging of the manipulated synapses. For this purpose, I use peroxidase-based genetic tags (HRP, APEX2, etc.) and correlative light electron microscopy (CLEM) to identify the synapses of interest. Combined with sparse transfection and non-invasive stimulation strategies, we investigate long-term (up to 3 days) synaptic changes. My aim is to establish a readout of synaptic plasticity at the ultrastructural level.
Rui Wang
The function of endoplasmic reticulum (ER) in spines
The history of activity of a neuron affects the calcium concentration inside the endoplasmic reticulum (ER). The ER is a complex organelle, distributed throughout neurons, including the soma, axon and dendritic arbor, and is present in some dendritic spines. While it is well known for its role in protein synthesis and transport, and for its ability to release calcium, less is known about how it affects synaptic plasticity. It has previously been shown that ER is stably present in 10% of dendritic spines while a further 60% are visited by transient ER every 5 h, with functional consequences for plasticity (Perez-Alvarez et al., Nat Commun. 2020). We have also previously shown that IP3R-mediated calcium release from the ER can have dramatic effects on synaptic plasticity (Holbro et al., PNAS 2009).
Using two-photon microscopy, we aim to investigate the role of the ER in dendritic spines, the conditions under which it acts as a calcium source or sink, and how this affects synaptic plasticity.
Kelsey Zingg
Automatizing image acquisition and analysis
Two-photon microscopy is the major analytical technique used in our lab and easily produces large amounts of data. I am working on establishing image processing workflows using both commercial software and custom code (Matlab, python) so that quantitative information can be extracted from the acquired images in a reproducible way (Laprell et al., 2021). I am also involved in modifying instrument software to add not-so-common features to the repertoire of experimental tools (Perez-Alvarez et al., 2020). To interpret experimental results, it is often useful to test your intuition with a biophysically realistic model. I contributed to the analysis of vesicular glutamate release at synapses by simulating the signaling event, using the Monte-Carlo simulation environment MCell (Dürst et al., 2020).
Christian Schulze
Representations of time and space in the hippocampal circuit
Neuronal activity patterns occur in a subset of neurons during encoding of memories. These ensembles are then reactivated when animals revisit the space in which the memory was encoded or when a particular (conditioned) stimulus, associated with that experience, is presented to the animals. We are interested in understanding how these patterns emerge from the vast number of neurons inside a mouse brain. Why these cells and not others? Is there a molecular or physiological signature that pre-selects them? In particular, we focus on the dentate gyrus (DG) of the hippocampus, which is a structure with extremely high neuronal density but with only a few neurons (<10%) active at a given time. The DG is fundamental for episodic memories and serves as a discriminator of similar experiences. We use mouse lines that have been genetically engineered to identify (tag) and exclusively express tools that allow their manipulation in neuronal ensembles. These tools are expressed only in active ensembles and in a chosen temporal window of experience. We challenge the mice to learn a particular escape location in a water tank (Morris water maze). Mice take several days to master this task, and we can monitor how learning affects the dynamics of active ensembles. Combining the selective tagging with opto- and chemogenetic tools, we can interrogate how the spatial information needed to solve the task is integrated over time in these neuronal ensembles. This project is funded by SFB936.
Andreas Franzelin & Lennart Beck
Collaborations
We collaborate with Anne Schäfer (NYC), Antoine Triller ) and Nils Brose to investigate the communication between microglia and neurons.
We collaborate with Peter Hegemann (Berlin) and Georg Nagel (Würzburg) to develop novel optogenetic tools.
We collaborate with Katalin Török (London) to develop fast genetically encoded sensors.
We collaborate with Rapp Optoelectronics (Wedel) to develop optical stimulation devices.
Behavioral experiments are performed with Fabio Morellini and Matthias Kneussel (ZMNH).
We collaborate with Tim Magnus to investigate synaptic plasticity in the recovery phase after stroke.
Dr. Shiqiang Gao (optogenetic tool development - with Peter Hegemann and Georg Nagel )
We collaborate with Christian Lohr to investigate the role of astrocytic cAMP on neuronal and synaptic function .