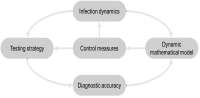
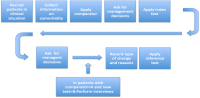
Statistical methods for diagnostic studies
An effective therapy requires a correct diagnosis. For this you need diagnostic tests which are evaluated in so-called diagnostic studies. The development of statistical methods for planning and analyzing diagnostic studies and diagnostic meta-analysis was neglected a long time. Therefore, we aim to close the still existing methodological gaps. Currently we are working on the development of flexible designs for diagnostic studies within the DFG-project entitled