Dissecting Sex Differences in the Immune Responses to Vaccines
Vaccination represents a major milestone in global public health, preventing millions of new infections and deaths worldwide each year. Sexual dimorphism in vaccine responses is increasingly recognised as a critical factor influencing immunogenicity and reactogenicity. For most licensed vaccines, cisgender females typically develop stronger antibody responses but also experience more adverse reactions than cisgender males. However, little is known about these differences following vector-based vaccines such as the modified vaccinia virus Ankara (MVA) vaccine platform.
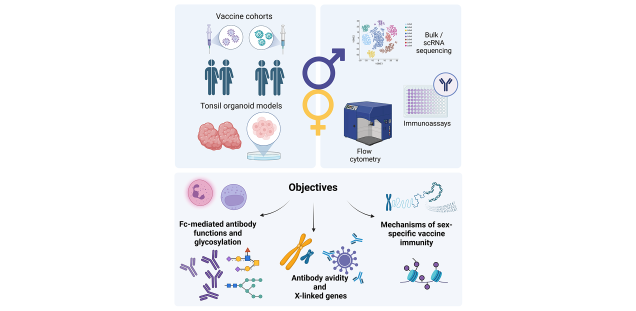
This project aims to investigate sex differences in response to MVA and influenza vaccines and better understand the underlying mechanisms. We will examine the contribution of non-neutralising Fc-mediated antibodies to differential vaccine immunogenicity and whether sex differences in Fc-mediated antibody functions, Ig isotypes and antibody glycosylation patterns contribute to differences in the vaccine response. We will also investigate whether sex-specific vaccine responses result from the enhanced expression of X-linked genes that escape X chromosome inactivation (XCI). Our hypotheses will be tested using data and biomaterials from longitudinal human vaccine cohorts, focusing on the MVA-MERS-S vaccine candidate. In addition, we will use an influenza vaccine cohort as a vaccine model for our research. Ex vivo human studies will be complemented by analyses of sex differences in the induction of B cells, plasmablasts and T follicular helper cells using tonsil organoid systems.
Understanding the mechanisms responsible for sex differences in vaccine-induced immune responses may allow strategic modulation of vaccine-induced immunity and help to develop more personalised vaccination strategies.
PROJECT-RELATED PUBLICATIONS
Ilka Grewe, Tamara Zoran, Marylyn Martina Addo (2026)
Sex Differences in Response to Viral Vector Vaccines-Implications for Future Vaccine Design.
Immunol Rev. 338(1):e70098
read more
Raadsen M, Dahlke C, Fathi A, Hardtke S, […], Sutter G, Becker S, Haagmans BL, and Addo MM and Group ‘MVA-MERS-S_DF-1 Study’. (2025)
The Safety, Immunogenicity, and Optimal Dosing of an MVA-Based Vaccine Against MERS Coronavirus in Healthy Adults: A Phase 1b, Randomised, Placebo-Controlled, Double-Blind Clinical Trial.
Lancet Infect Dis I 25:231-242
Mayer L, Weskamm LM, Fathi A, Kono M, Heidepriem J, Krähling V, Mellinghoff SC, Ly ML, Friedrich M, Hardtke S, Borregaard S, Hesterkamp T, Loeffler FF, Volz A, Sutter G, Becker S, Dahlke C, Addo MM. (2024)
MVA-based vaccine candidates encoding the native or prefusion-stabilized SARS-CoV-2 spike reveal differential immunogenicity in humans.
NPJ Vaccines I 9:20
Beisel C, Jordan-Paiz A, Köllmann S, Ahrenstorf AE, Padoan B, Barkhausen T, Addo MM, Altfeld M. (2023)
Sex differences in the percentage of IRF5 positive B cells are associated with higher production of TNF-α in women in response to TLR9 in humans.
Biology of Sex Differences I 14:11
read more
Anahita Fathi, Marylyn M Addo, Christine Dahlke. (2021)
Sex Differences in Immunity: Implications for the Development of Novel Vaccines Against Emerging Pathogens.
Front Immunol I 11:601170
read more
Koch T, Dahlke C, Fathi A, Kupke A, Krähling V, Okba MA, Halwe S, Rohde C, Eickmann M, Volz A, Hesterkamp T, Jambrecina A, Borregaard S, Ly ML, Zinser ME, Bartels E, Poetsch J, Neumann R, Fux R, Schmiedel S, Lohse AW, Haagmans BL, Sutter G, Becker S, Addo MM. (2020)
Safety and immunogenicity of a modified vaccinia virus Ankara vector vaccine candidate for Middle East respiratory syndrome: an open-label, phase 1 trial.
Lancet Infect Dis I 20:827-838
read more
Heidepriem J, Krähling V, Dahlke C, Wolf T, Klein F, Addo MM*, Becker S*, Loeffler F*. (2020)
Epitopes of naturally acquired and vaccine-induced anti-Ebola virus glycoprotein antibodies in single amino acid resolution.
Biotechnol J I 15:e2000069
read more
Friedrich SK, Schmitz R, Bergerhausen M, Lang J, Cham LB, Duhan V, Häussinger D, Hardt C, Addo M, Prinz M, Asano K, Lang PA, Lang KS. (2020)
Usp18 Expression in CD169+ Macrophages is Important for Strong Immune Response after Vaccination with VSV-EBOV.
Vaccines (Basel) I 8:E142
read more
Ehrhardt S A, Zehner M, Krähling V, Cohen-Dvashi H, Kreer C, Elad N, Gruell H, Ercanoglu MS, Schommers P, Gieselmann L, Eggeling R, Dahlke C, Wolf T, Pfeifer N, Addo, MM, Diskin R, Becker S, & Klein F. (2019)
Polyclonal and convergent antibody response to Ebola virus vaccine rVSV-ZEBOV.
Nature medicine I 25:1589–1600
read more
Poetsch J.H., Dahlke C, Zinser ME, Kasonta R, Lunemann S, Rechtien A, My Linh Ly, Stubbe H.C., Krähling V, Biedenkopf N, Eickmann M, Fehling SK, Olearo F, Strecker T, Sharma P, Lang KS, Lohse AW, Schmiedel S, Becker S, VSV-Ebola Consortium (VEBCON), Addo MM. (2019)
Detectable Vesicular Stomatitis Virus (VSV)–Specific Humoral and Cellular Immune Responses Following VSV– Ebola Virus Vaccination in Humans.
J Infect Dis I 219:556-561
read more
Rechtien A, Richert L, Lorenzo H, Martrus G, Hejblum B, Dahlke C, Kasonta R, Zinser M, Stubbe H, Matschl U, Lohse A, Krähling V, Eickmann M, Becker S; VEBCON Consortium, Thiébaut R, Altfeld M, Addo MM. (2017)
Systems Vaccinology Identifies an Early Innate Immune Signature as a Correlate of Antibody Responses to the Ebola Vaccine rVSV-ZEBOV.
Cell Rep I 20:2251-2261
read more
Ruibal P, Oestereich L, Lüdtke A, Becker-Ziaja B, Wozniak DM, Kerber R, Herker E, Jacobs T, Kretschmer B, Severi E, Ouedraogo N, Lago M, Negredo A, Franco L, Anda P, Schmiedel S, Kreuels B, Wichmann D, Addo MM, Lohse AW, De Clerck H, Nanclares C, Jonckheere S, Van Herp M, Sprecher A, Xiaojiang G, Carrington M, Miranda O, Castro CM, Gabriel M, Drury P, Formenty P, Diallo B, Koivogui L, Magasouba N, Carroll MW, Günther S, Muñoz-Fontela C. (2016)
Unique human immune signature of Ebola virus disease in Guinea.
Nature I 533:100-4
read more
Dahlke C, Kasonta R, Lunemann S, Krähling V, Zinser ME, Biedenkopf N, Fehling SK, Ly ML, Rechtien A, Stubbe HC, Olearo F, Borregaard S, Jambrecina A, Stahl F, Strecker T, Eickmann M, Lütgehetmann M, Spohn M, Schmiedel S, Lohse AW, Becker S, Addo MM, VEBCON Consortium. (2017)
Dose-dependent T-cell Dynamics and Cytokine Cascade Following rVSV-ZEBOV Immunization.
EBioMedicine I 19:107-11
read more
Agnandji ST, Huttner A, Zinser ME, Njuguna P, Dahlke C, Fernandes JF, Yerly S, Dayer JA, Kraehling V, Kasonta R, Adegnika AA, Altfeld M, Auderset F, Bache EB, Biedenkopf N, Borregaard S, Brosnahan JS, Burrow R, Combescure C, Desmeules J, Eickmann M, Fehling SK, Finckh A, Goncalves AR, Grobusch MP, Hooper J, Jambrecina A, Kabwende AL, Kaya G, Kimani D, Lell B, Lemaître B, Lohse AW, Massinga-Loembe M, Matthey A, Mordmüller B, Nolting A, Ogwang C, Ramharter M, Schmidt-Chanasit J, Schmiedel S, Silvera P, Stahl FR, Staines HM, Strecker T, Stubbe HC, Tsofa B, Zaki S, Fast P, Moorthy V, Kaiser L, Krishna S, Becker S, Kieny MP, Bejon P*, Kremsner PG*, Addo MM*, Siegrist CA*. (2016)
Phase 1 Trials of rVSV Ebola Vaccine in Africa and Europe.
N Engl J Med I 374:1647-60
read more
Addo MM, Altfeld M. (2014)
Sex-based differences in HIV type 1 pathogenesis.
J Infect Dis I 209 Suppl 3:S86-92
read more
* equal contribution