Sex-specific responses of myeloid cells to infectious challenges
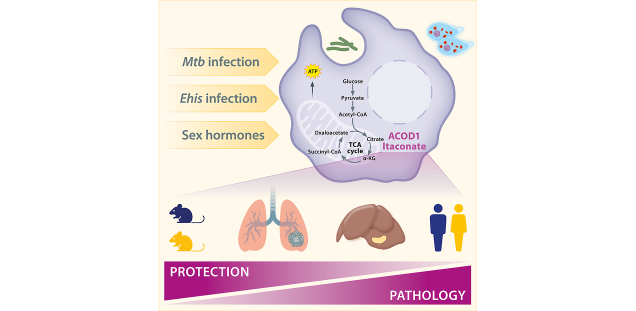
Immune responses to foreign antigens are often influenced by sex-specific biological factors. Infections with the protozoan parasite Entamoeba histolytica (Ehis) and the bacterium Mycobacterium tuberculosis (Mtb) are more common in adult men. In a murine model of hepatic amoebiasis, classical monocytes, resident macrophages, and neutrophils contribute to liver damage following parasite invasion by producing high levels of pro-inflammatory mediators like TNF and chemokines. Our previous studies in mice and human cells showed that testosterone, typically linked to immune suppression, plays a key role in promoting the production of these pro-inflammatory mediators in monocytes. Monocytes also seem to play a role in the sex-specific disparity in lung pathology during Mtb infection. In both the peripheral circulation and lung tissue, the presence of moncytes is notably higher in males compared to females in the animal model of the disease. Moreover, upon infiltration of lung tissue, monocytes demonstrate increased expression of specific inflammatory markers, implying their participation in immunopathological processes similar to those observed in amoebiasis. Single-cell analysis of monocytes from transgender men undergoing testosterone-based gender-affirming hormone therapy (GAHT) confirmed that testosterone enhances their pro-inflammatory state. Interestingly, we also observed an increase in aconitate decarboxylase 1 (ACOD1), a key immunometabolism regulator, suggesting the activation of a testosterone-mediated mechanism that may counterbalance this pro-inflammatory response.
We hypothesize that immune metabolism plays a key role in modulating monocyte-dependent immunopathology and antimicrobial responses, and aim to explore the interaction between sex hormones and immunometabolism in myeloid cells during Ehis and Mtb infection.
We will study the sex-specific interplay of the immune metabolism with the immune response of myeloid cells and its impact on the outcome of infection.
PROJECT-RELATED PUBLICATIONS
Charlotte Sophie Hansen, Julie Sellau, Anastasia Langanz, Leonie Marie Weskamm, Nele Wichern, Annika Bea, Johannes Brandi, Helena Fehling, Nils Groth, Melanie Lütkemeyer, Matthias Knödler, Henrik Nausch, Eirini Stivachti, Barbara Honecker, Hanna Lotter (2026)
Sex shapes CD64 expression and vaccine-induced monocytic responses.
Biol Sex Differ. 17(1):77
read more
Barbara Honecker, Charlotte Sophie Hansen, Hanna Lotter (2026)
Sex in Immune Cells and Parasitic Diseases – A Complex Relationship.
Immunol Rev. 337(1):e70097
read more
Annika Bea, Helena Fehling, Fabian Hausmann, Fahten Margot Habib, Melanie Lütkemeyer, Lara Buer, Carola Schäfer, Charlotte Sophie Hansen, Barbara Honecker, Stefan Bonn, Bianca Elisabeth Schneider, Joachim Clos, Hanna Lotter (2025)
Transcriptome profiling of L. infantum-infected human macrophages reveals sex-specific type I interferon induction
PLoS Pathog 21:e1013427.
read more
Barbara Honecker, Valentin A Bärreiter, Katharina Höhn, Balázs Horváth, Karel Harant, Nahla Galal Metwally, Claudia Marggraff, Juliett Anders, Stephanie Leyk, Maria Del Pilar Martínez-Tauler, Annika Bea, Charlotte Hansen, Helena Fehling, Melanie Lütkemeyer, Stephan Lorenzen, Sören Franzenburg, Hanna Lotter, Iris Bruchhaus (2025)
Entamoeba histolytica extracellular vesicles drive pro-inflammatory monocyte signaling
PLoS Negl Trop Dis 19:e0012997
read more
Hertz D, Eggers L, von Borstel L, Goldmann T, Lotter H, Schneider BE. (2025)
Turning the Tables: Loss of Adaptive Immunity Reverses Sex Differences in Tuberculosis.
Immuno.I 5:4.
read more
Sellau J, Hansen CS, Gálvez RI, Linnemann L, Honecker B, Lotter H. (2024)
Immunological clues to sex differences in parasitic diseases.
Trends Parasitol I 40:1029-1041
read more
Marco Er-Lukowiak, Sonja Hänzelmann, Moritz Rothe, David T Moamenpour, Fabian Hausmann, Robin Khatri, Charlotte Hansen, Jennifer Boldt, Valentin A Bärreiter, Barbara Honecker, Annika Bea, Marie Groneberg, Helena Fehling, Claudia Marggraff, Dániel Cadar, Stefan Bonn, Julie Sellau, Hanna Lotter (2023)
Testosterone affects type I/type II interferon response of neutrophils during hepatic amebiasis
Front Immunol. 14:1279245
read more
Groneberg M, Hoenow S, Marggraff C, Fehling H, Metwally NG, Hansen C, Bruchhaus I, Tiegs G, Sellau J, Lotter H. (2021)
HIF-1α modulates sex-specific Th17/Treg responses during hepatic amoebiasis
J Hepatol. S0168-8278(21)02082-1. Online ahead of print.
read more
Sellau J, Groneberg M, Hoenow S, Lotter H. (2021)
The underlying cellular immune pathology of Entamoeba histolytica-induced hepatic amoebiasis
J Hepatol 75:481-482
read more
Sellau J, Groneberg M, Fehling H, Thye T, Hoenow S, Marggraff C, Weskamm M, Hansen C, Stanelle-Bertram S, Kuehl S, Noll J, Wolf V, Metwally NG, Hagen SH, Dorn C, Wernecke J, Ittrich H, Tannich E, Jacobs T, Bruchhaus I, Altfeld M, Lotter H. (2020)
Androgens predispose males to monocyte-mediated immunopathology by inducing the expression of leukocyte recruitment factor CXCL1.
Nat Commun I 11:3459
read more
Hertz D, Dibbern J, Eggers L, von Borstel L, Schneider BE. (2020)
Increased male susceptibility to Mycobacterium tuberculosis infection is associated with smaller B cell follicles in the lungs.
Scientific Reports I 10: 5142
read me
Dibbern J, Eggers L, Schneider BE. (2017)
Sex differences in the C57BL/6 model of Mycobacterium tuberculosis infection.
Scientific Reports I 7: 10957
read more