 Research at the UKE?
Research at the UKE?

Institute of Immunology
The Institute of Immunology is responsible for the University Hospital’s routine laboratory diagnosis of immune-mediated diseases, and for academic teaching and training of students in immunology. Research is a core pillar of our Institute, dedicated to investigating aberrant pathogenic and regulatory immune mechanisms that underlie disease.
Recent advances in our understanding of such mechanisms have led to a growing number of clinical interventions aimed at therapeutically modulating both innate and adaptive immune responses in immune-mediated conditions.

“The outcome of many infections differ between females and males, but the underlying immune mechanisms are incompletely understood. We investigate the impact of sex steroid hormones and genes encoded by the X and Y chromosome on antiviral immunity, and the consequences for viral pathogenesis.”
Director of the institute – Institute of Immunology

“T cells play a central role in immune responses against pathogens, but they can also cause autoimmunity and immunopathology. Therefore, understanding how T cells are regulated in health and disease is crucial for developing novel, targeted therapies to control infections and prevent immune-mediated disorders.”

“Nanobodies and antibodies are essential research tools for understanding the immune system. They also hold promise as novel therapeutics for immune mediated diseases.”
Research Group Leader

“'Nanobodies are the small, single variable domains of camelid heavy-chain antibodies that can readily be incorporated into the capsid of AAV gene therapy vectors. Insertion of a membrane protein-specific nanobody targets the AAV to cells expressing the respective membrane protein, thereby improving therapeutic specificity and efficiency.”

“Betaherpesviruses are species-specific opportunistic pathogens that have co-evolved with their hosts. We investigate how virus-encoded immune evasins influence host immunity and shape the clinical outcome of infection.”
Research Group Leader

“Identifying the factors and mechanisms driving early immune deviation is essential for informing smarter strategies to prevent immune-mediated diseases.”
Current projects
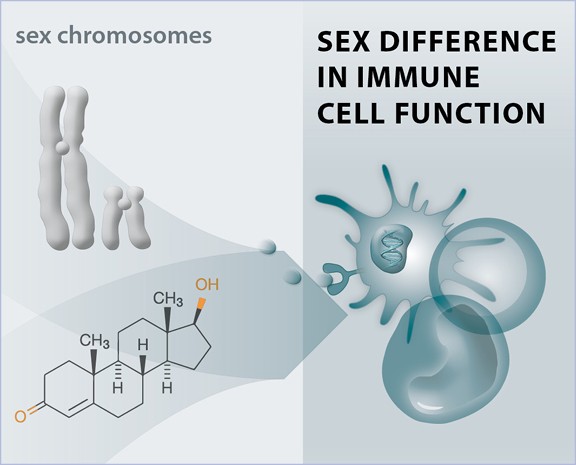
Sex differences in antiviral immunity
The clinical manifestations of viral infections differ between women and men. In most viral infections, including HIV-1, HCV and SARS-CoV-2 infections, females exhibit better control of viral replication and milder disease manifestations in acute infections. However, long-term consequences of viral infections sometimes are more severe in females, as reflected by faster HIV-1 disease progression to AIDS for the same level of viral replication and higher incidences of Long COVID. We investigate the mechanisms underlying these sex differences in the manifestations of viral infections, with a particular focus on the role of antiviral Type I IFN responses and NK cell responses.
This project is embedded in the
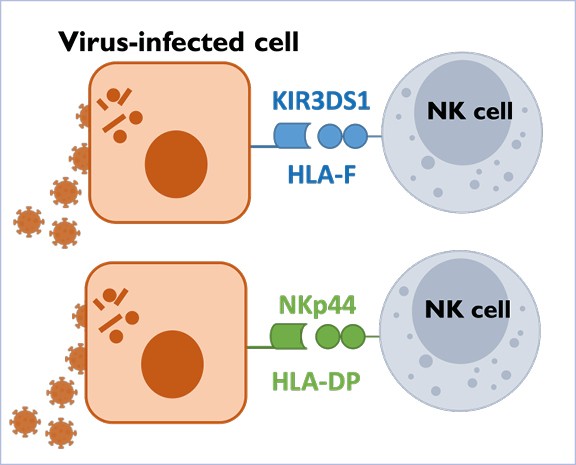
Antiviral NK cell responses
NK cells play a central role in the control of viral infections, but the precise mechanisms that allow NK cells to recognize and kill virus-infected cells remain incompletely understood. The Altfeld Lab investigates the receptor-ligand interactions that enable human NK cells to recognize virus-infected cells, with a particular focus on the role of Killer cell Immunoglobulin-like Receptors (KIRs) and Natural Cytotoxicity Receptors (NCRs).
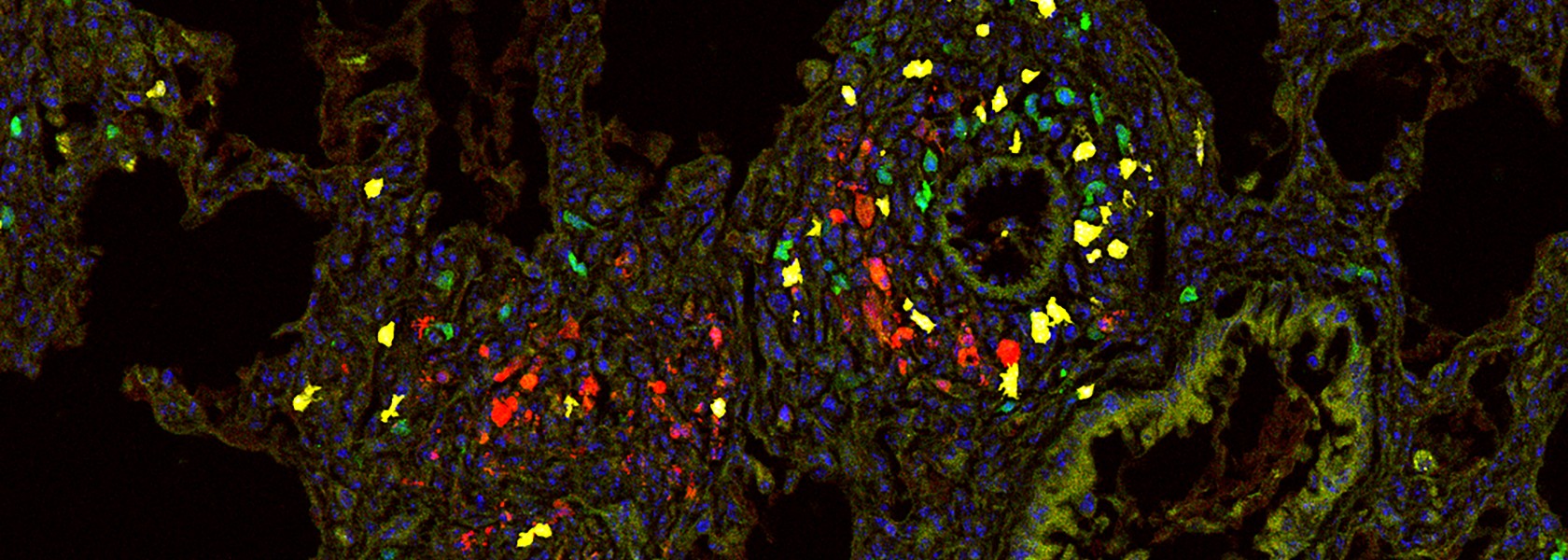
Tissue-resident memory T cells
Tissue-resident memory T (TRM) cells are a distinct subset of memory T cells mainly located at barrier sites such as the skin and mucosal tissues. They act as local sentinels for previously encountered pathogens, providing immediate protection against reinfection. In contrast, their role in autoimmunity and tissue regeneration is less well understood.
Our research focuses on the role of TRM cells in the development and progression of glomerulonephritis, with a particular focus on the mechanisms of their activation, how they interact with other renal cells, and their contribution to renal damage. A second project focuses on the function of TRM cells in the female reproductive tract, how they adapt to tissue remodeling during pregnancy, and their contribution to fetal tolerance.
Learn more
Learn more
Regulation of T cells in bacterial infection and autoimmunity
The precise and efficient function of the adaptive immune system depends on the regulation of T cells. We are investigating several aspects of T cell regulation. T cells express proteases from the A Disintegrin and Metalloproteinase (ADAM) family on their surface. Our focus is on the function of ADAM10 on T cells, which sheds the extracellular domains of surface proteins, including adhesion proteins and cytokine receptors. This interferes with cell-to-cell interaction and cytokine signaling. The transcription factor Interferon Regulatory Factor 4 (IRF4) is essential for the activation and differentiation of lymphocytes. We are investigating the function of IRF4 in different effector and memory T cell subsets. An increase in the cytoplasmic Ca²⁺ concentration is a critical consequence of T cell receptor stimulation. We analyze the role of different adenine nucleotide-targeted Ca²⁺ channels in this process, as well as the consequences of their activation for T cell differentiation and function.
Learn more
Modulating adenine nucleotide signaling with antibodies
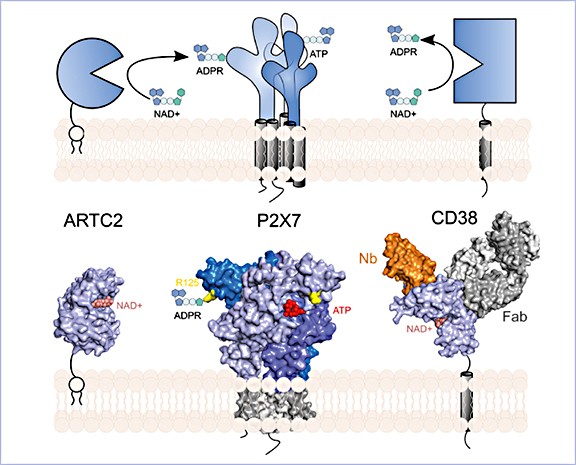
The goal of the project is to generate and develop antibodies and nanobodies against the key players of adenine nucleotide signaling, e.g. ectoenzymes (CD38, CD39, CD73, CD203) and nucleotide-gated ion channels (P2X4, P2X5, P2X7) – as tools for basic research and potentially as new therapeutics for auto-immune and chronic inflammatory diseases.
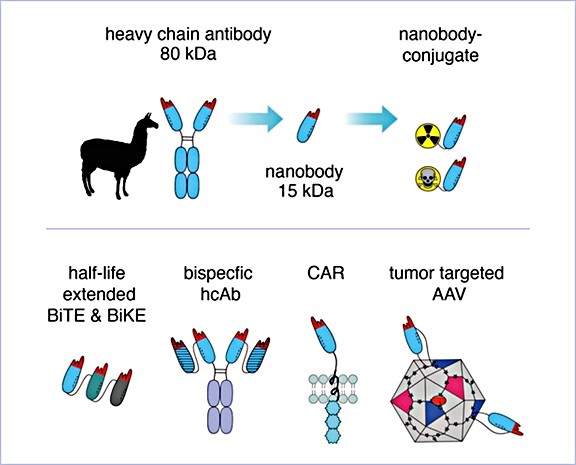
THUNDER – A nanobody pipeline for oncology
The goal of the project is to develop nanobody-based therapeutics that target membrane proteins on tumor cells and mediators of an immunosuppressive tumor micro-environment.
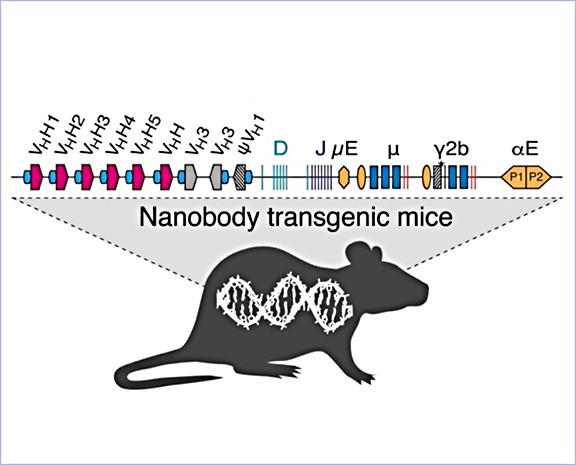
Nanobodies from LamaMice
The goal of the project is to generate and characterize nanobodies as tools for research and therapy with collaborators at the UKE in the fields of biochemistry, immunity, oncology, nephrology and neurology. The project also aims to make UKE-proprietary LamaMice accessible to others via the contract research organizations Genovac and preclinics.
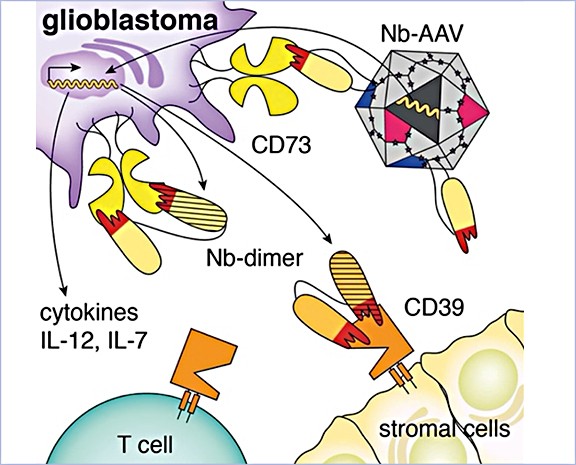
Targeting of AAV gene therapy vectors with membrane protein-specific nanobodies
The project focuses on using membrane protein-specific nanobodies to target AAV gene therapy vectors to specific cell types. The goal is to develop improved and efficient vectors for the treatment of glioblastoma brain tumors and muscular dystrophies.
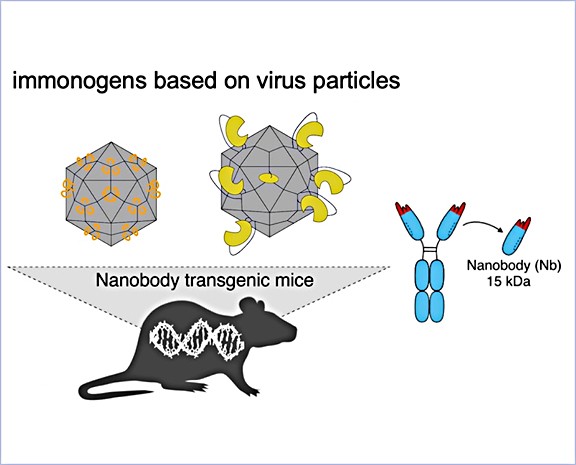
Development of chimeric virus-like particles as potent immunogens
The goal of the project is to develop chimeric virus-like particles based on AAV vectors as potent immunogens for the generation of nanobodies in the UKE-proprietary LamaMice. These LamaMice produce the special heavy-chain-only antibodies from llamas upon immunization.
Host factors in betaherpesvirus immunity
Betaherpesviruses are opportunistic pathogens whose clinical impact is shaped by the host’s immune response. This project seeks to identify key factors - including age, sex, and tissue-specific immunity - that determine effective protection against these viruses.
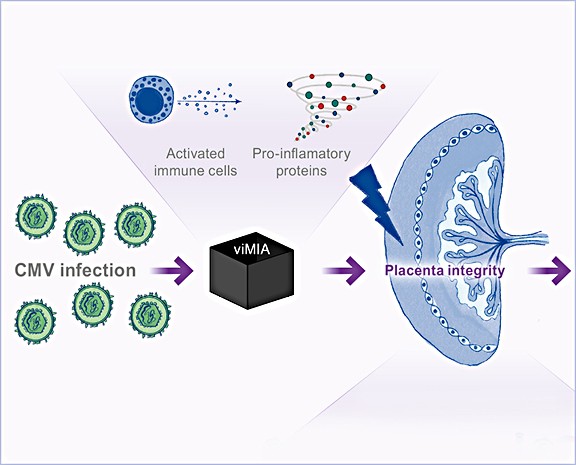
Virus-induced maternal immune activation infection
Maternal infection with cytomegalovirus causes maternal immune activation, and the virus can cross the placenta, leading to congenital infection. This project aims to understand the impact of virus-induced maternal immune activation on maternal and fetal health.
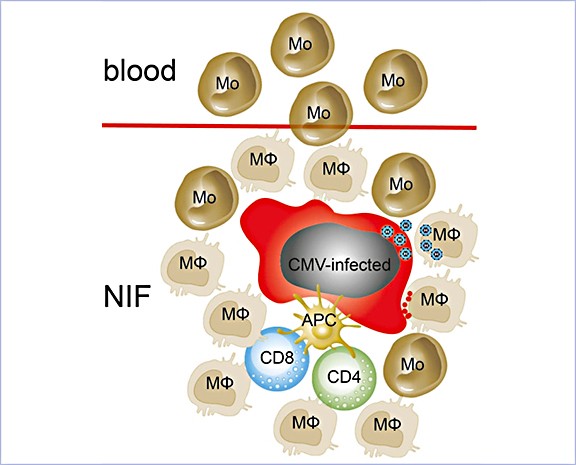
Role of myeloid cells in early life cytomegalovirus infection
This project aims to characterize myeloid cell phenotypes after CMV infection and to define their functional roles in the antiviral response. It also seeks to identify and modify key molecules that govern myeloid cell-mediated immunity.
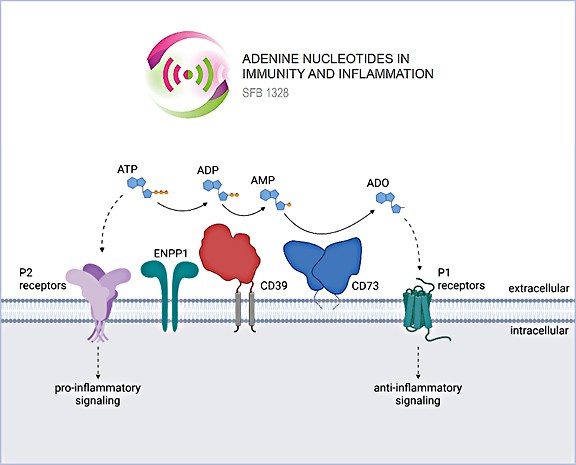
Adenine Nucleotides and Immune Regulation
Extracellular ATP levels increase during cellular stress or inflammation, triggering a pro-inflammatory response. Ectonucleotidases sequentially degrade ATP to immune-modulatory adenosine. We study the mechanisms regulating the bioavailability of ATP and adenosine, and how purinergic signaling shapes immune responses in health and disease.
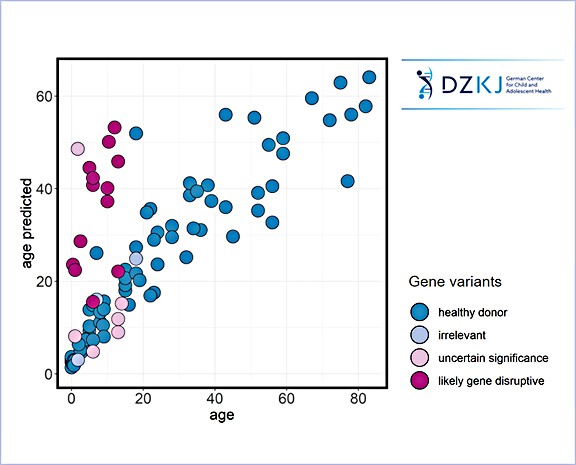
Early deviation in human T cell development
Inborn errors of immunity or a bumpy start in life can disrupt T cell development. We use multiomic approaches in combination with thymus organoids to study factors that influence T cell development. Machine learning tools help us to define ‘normality’ in the T cell compartment over a life time and to predict early deviation.

Immune profiling of disease cohorts
The number of health conditions with a recognized immune component is increasing, as is the use of immune-modulatory and enzyme-replacement therapies. We specialize in flow cytometry–based and targeted proteomics approaches to immune profile the human immune system across pediatric and adult disease cohorts, aiming to identify endotypes that can inform more personalized treatment strategies.
Team – Altfeld


Prof. Dr. med. Marcus Altfeld
Director of the institute – Institute of Immunology




Publications Altfeld Lab
Zecher BF, Ellinghaus D, Schloer S, Niehrs A, Padoan B, Baumdick ME, Yuki Y, Martin MP, Glow D, Schröder-Schwarz J, Niersch J, Brias S, Müller LM, Habermann R, Kretschmer P, Früh T, Dänekas J, Wehmeyer MH, Poch T, Sebode M; International PSC Study Group (IPSCSG); Ellinghaus E, Degenhardt F, Körner C, Hoelzemer A, Fehse B, Oldhafer KJ, Schumacher U, Sauter G, Carrington M, Franke A, Bunders MJ, Schramm C, Altfeld M. Gut. 2024 Jan 5;73(2):325-337.
Abstract
Objective: Primary sclerosing cholangitis (PSC) is characterised by bile duct strictures and progressive liver disease, eventually requiring liver transplantation. Although the pathogenesis of PSC remains incompletely understood, strong associations with HLA-class II haplotypes have been described. As specific HLA-DP molecules can bind the activating NK-cell receptor NKp44, we investigated the role of HLA-DP/NKp44-interactions in PSC.
Design: Liver tissue, intrahepatic and peripheral blood lymphocytes of individuals with PSC and control individuals were characterised using flow cytometry, immunohistochemical and immunofluorescence analyses. HLA-DPA1 and HLA-DPB1 imputation and association analyses were performed in 3408 individuals with PSC and 34 213 controls. NK cell activation on NKp44/HLA-DP interactions was assessed in vitro using plate-bound HLA-DP molecules and HLA-DPB wildtype versus knock-out human cholangiocyte organoids.
Results: NKp44+NK cells were enriched in livers, and intrahepatic bile ducts of individuals with PSC showed higher expression of HLA-DP. HLA-DP haplotype analysis revealed a highly elevated PSC risk for HLA-DPA1*02:01~B1*01:01 (OR 1.99, p=6.7×10-50). Primary NKp44+NK cells exhibited significantly higher degranulation in response to plate-bound HLA-DPA1*02:01-DPB1*01:01 compared with control HLA-DP molecules, which were inhibited by anti-NKp44-blocking. Human cholangiocyte organoids expressing HLA-DPA1*02:01-DPB1*01:01 after IFN-γ-exposure demonstrated significantly increased binding to NKp44-Fc constructs compared with unstimulated controls. Importantly, HLA-DPA1*02:01-DPB1*01:01-expressing organoids increased degranulation of NKp44+NK cells compared with HLA-DPB1-KO organoids.
Conclusion: Our studies identify a novel PSC risk haplotype HLA-DP A1*02:01~DPB1*01:01 and provide clinical and functional data implicating NKp44+NK cells that recognise HLA-DPA1*02:01-DPB1*01:01 expressed on cholangiocytes in PSC pathogenesis.
Niehrs A, Garcia-Beltran WF, Norman PJ, Watson GM, Hölzemer A, Chapel A, Richert L, Pommerening-Röser A, Körner C, Ozawa M, Martrus G, Rossjohn J, Lee JH, Berry R, Carrington M, Altfeld M. Nat Immunol. 2019 Sep;20(9):1129-1137.
Abstract
Natural killer (NK) cells can recognize virus-infected and stressed cells using activating and inhibitory receptors, many of which interact with HLA class I. Although early studies also suggested a functional impact of HLA class II on NK cell activity the NK cell receptors that specifically recognize HLA class II molecules have never been identified. We investigated whether two major families of NK cell receptors, killer-cell immunoglobulin-like receptors (KIRs) and natural cytotoxicity receptors (NCRs), contained receptors that bound to HLA class II, and identified a direct interaction between the NK cell receptor NKp44 and a subset of HLA-DP molecules, including HLA-DP401, one of the most frequent class II allotypes in white populations. Using NKp44ζ+ reporter cells and primary human NKp44+ NK cells, we demonstrated that interactions between NKp44 and HLA-DP401 trigger functional NK cell responses. This interaction between a subset of HLA-DP molecules and NKp44 implicates HLA class II as a component of the innate immune response, much like HLA class I. It also provides a potential mechanism for the described associations between HLA-DP subtypes and several disease outcomes, including hepatitis B virus infection, graft-versus-host disease and inflammatory bowel disease.
Team – Mittrücker


Prof. Dr. rer. nat. Hans-Willi Mittrücker
Research Group Leader – Infection Biology






Publications Mittrücker Lab
Asada N, Ginsberg P, Paust HJ, Song N, Riedel JH, Turner JE, Peters A, Kaffke A, Engesser J, Wang H, Zhao Y, Khatri R, Gild P, Dahlem R, Diercks BP, Das S, Ignatova Z, Huber TB, Prinz I, Gagliani N, Mittrücker H-W*, Krebs CF*, Panzer U*. Nat Immunol. 2025 26:557-566.
Abstract
Tissue-resident memory T (TRM) cells are a specialized T cell population that reside in tissues and provide a rapid protective response upon activation. Here, we showed that human and mouse CD4+TRM cells existed in a poised state and stored messenger RNAs encoding proinflammatory cytokines without protein production. At steady state, cytokine mRNA translation in TRM cells was suppressed by the integrated stress response (ISR) pathway. Upon activation, the central ISR regulator, eIF2α, was dephosphorylated and stored cytokine mRNA was translated for immediate cytokine production. Genetic or pharmacological activation of the ISR-eIF2α pathway reduced cytokine production and ameliorated autoimmune kidney disease in mice. Consistent with these results, the ISR pathway in CD4+ TRM cells was downregulated in patients with immune-mediated diseases of the kidney and the intestine compared to healthy controls. Our results indicated that stored cytokine mRNA and translational regulation in CD4+ TRM cells facilitate rapid cytokine production during local immune response.
* shared authorship
Bertram T, Reimers D, Lory NC, Schmidt C, Schmid J, C Heinig L, Bradtke P, Rattay G, Zielinski S, Hellmig M, Bartsch P, Rohde H, Nuñez S, Rosemblatt MV, Bono MR, Gagliani N, Sandrock I, Panzer U, Krebs CF, Meyer-Schwesinger C, Prinz I, Mittrücker H-W. Proc Natl Acad Sci U S A. 2023 120:e2210490120.
Abstract
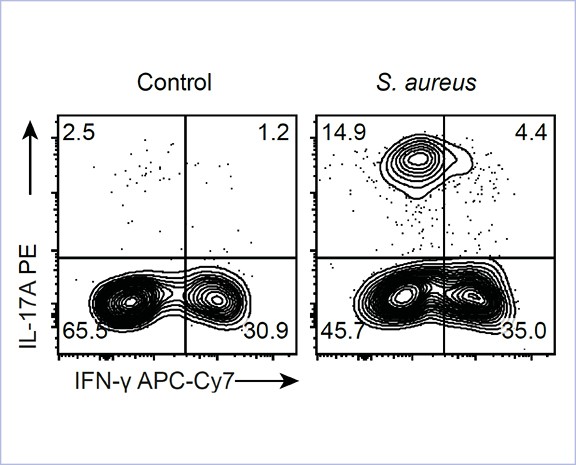
γδ T cells are involved in the control of Staphylococcus aureus infection, but their importance in protection compared to other T cells is unclear. We used a mouse model of systemic S. aureusinfection associated with high bacterial load and persistence in the kidney. Infection caused fulminant accumulation of γδ T cells in the kidney. Renal γδ T cells acquired tissue residency and were maintained in high numbers during chronic infection. At day 7, up to 50% of renal γδ T cells produced IL-17A in situ and a large fraction of renal γδ T cells remained IL-17A+ during chronic infection. Controlled depletion revealed that γδ T cells restricted renal S. aureus replication in the acute infection and provided protection during chronic renal infection and upon reinfection. Our results demonstrate that kidney-resident γδ T cells are nonredundant in limiting local S. aureusgrowth during chronic infection and provide enhanced protection against reinfection.
* shared authorship
Harberts A, Schmidt C, Schmid J, Reimers D, Koch-Nolte F, Mittrücker H-W*, Raczkowski F*. Proc Natl Acad Sci U S A. 2021. 118:e2014553118.
Abstract
The transcription factor IRF4 is required for CD8+ T cell activation, proliferation, and differentiation to effector cells and thus is essential for robust CD8+ T cell responses. The function of IRF4 in memory CD8+ T cells yet needs to be explored. To investigate the role of IRF4 for maintaining differentiation state and survival of CD8+ memory T cells, we used a mouse model with tamoxifen-inducible Irf4 knockout to preclude effects due to inefficient memory cell differentiation in absence of IRF4. We infected mice with ovalbumin-recombinant listeria and induced Irf4 knockout after clearance of the pathogen. Loss of IRF4 resulted in phenotypical changes of CD8+ memory T cells but did not cause a reduction of the total memory T cell population. However, upon reencounter of the pathogen, CD8+ memory T cells showed impaired expansion and acquisition of effector functions. When compared to CD8+ effector memory T cells, CD8+ tissue-resident memory T cells (TRM cells) expressed higher IRF4 levels. Mice with constitutive Irf4 knockout had diminished CD8+TRM-cell populations, and tamoxifen-induced Irf4 deletion caused a reduction of this cell population. In conclusion, our results demonstrate that IRF4 is required for effective reactivation but not for general survival of CD8+ memory T cells. Formation and maintenance of CD8+ TRM cells, in contrast, appear to depend on IRF4.
* shared authorship
Krebs CF, Reimers D, Zhao Y, Paust HJ, Bartsch P, Nuñez S, Rosemblatt MV, Hellmig M, Kilian C, Borchers A, Enk LUB, Zinke M, Becker M, Schmid J, Klinge S, Wong MN, Puelles VG, Schmidt C, Bertram T, Stumpf N, Hoxha E, Meyer-Schwesinger C, Lindenmeyer MT, Cohen CD, Rink M, Kurts C, Franzenburg S, Koch-Nolte F, Turner JE, Riedel JH, Huber S, Gagliani N, Huber TB, Wiech T, Rohde H, Bono MR, Bonn S, Panzer U*, Mittrücker H-W*. Sci Immunol. 2020. 5:eaba4163.
Abstract
Although it is well established that microbial infections predispose to autoimmune diseases, the underlying mechanisms remain poorly understood. After infection, tissue-resident memory T (TRM) cells persist in peripheral organs and provide immune protection against reinfection. However, whether TRM cells participate in responses unrelated to the primary infection, such as autoimmune inflammation, is unknown. By using high-dimensional single-cell analysis, we identified CD4+ TRM cells with a TH17 signature (termed TRM17 cells) in kidneys of patients with ANCA-associated glomerulonephritis. Experimental models demonstrated that renal TRM17 cells were induced by pathogens infecting the kidney, such as Staphylococcus aureus, Candida albicans, and uropathogenic Escherichia coli, and persisted after the clearance of infections. Upon induction of experimental glomerulonephritis, these kidney TRM17 cells rapidly responded to local proinflammatory cytokines by producing IL-17A and thereby exacerbate renal pathology. Thus, our data show that pathogen-induced TRM17 cells have a previously unrecognized function in aggravating autoimmune disease.
* shared authorship
Team – Nolte


Prof. Dr. med. Friedrich Nolte
Research Group Leader





Team – Mann


Dr. Anna Marei Mann
Junior Research Group Leader





Publications Nolte Lab
Eden T, Schaffrath AZ, Wesolowski J, Stahler T, Tode N, Richter N, Schafer W, Hambach J, Hermans-Borgmeyer I, Woens J, Le Gall CM, Wendler S, Linke-Winnebeck C, Stobbe M, Budnicki I, Wanney A, Heitz Y, Schimmelpfennig L, Schweitzer L, Zimmer D, Stahl E, Seyfried F, Gebhardt AJ, Dieckow L, Riecken K, Fehse B, Bannas P, Magnus T, Verdoes M, Figdor CG, Hartlepp KF, Schleer H, Funer J, Tomas NM, Haag F, Rissiek B, Mann AM, Menzel S & Koch-Nolte F (2024). Nat Commun, 15: 4728.
Abstract
Due to their exceptional solubility and stability, nanobodies have emerged as powerful building blocks for research tools and therapeutics. However, their generation in llamas is cumbersome and costly. Here, by inserting an engineered llama immunoglobulin heavy chain (IgH) locus into IgH-deficient mice, we generate a transgenic mouse line, which we refer to as 'LamaMouse'. We demonstrate that LamaMice solely express llama IgH molecules without association to Igκ or λ light chains. Immunization of LamaMice with AAV8, the receptor-binding domain of the SARS-CoV-2 spike protein, IgE, IgG2c, and CLEC9A enabled us to readily select respective target-specific nanobodies using classical hybridoma and phage display technologies, single B cell screening, and direct cloning of the nanobody-repertoire into a mammalian expression vector. Our work shows that the LamaMouse represents a flexible and broadly applicable platform for a facilitated selection of target-specific nanobodies.
Eichhoff AM, Borner K, Albrecht B, Schafer W, Baum N, Haag F, Korbelin J, Trepel M, Braren I, Grimm D, Adriouch S & Koch-Nolte F (2019). Mol Ther Methods Clin Dev, 15:211-220.
Abstract
A limiting factor for the use of adeno-associated viruses (AAVs) as vectors in gene therapy is the broad tropism of AAV serotypes, i.e., the parallel infection of several cell types. Nanobodies are single immunoglobulin variable domains from heavy chain antibodies that naturally occur in camelids. Their small size and high solubility allow easy reformatting into fusion proteins. Herein we show that a membrane protein-specific nanobody can be inserted into a surface loop of the VP1 capsid protein of AAV2. Using three structurally distinct membrane proteins-a multispan ion channel, a single-span transmembrane protein, and a glycosylphosphatidylinositol (GPI)-anchored ectoenzyme-we show that this strategy can dramatically enhance the transduction of specific target cells by recombinant AAV2. Moreover, we show that the nanobody-VP1 fusion of AAV2 can be incorporated into the capsids of AAV1, AAV8, and AAV9 and thereby effectively redirect the target specificity of other AAV serotypes. Nanobody-mediated targeting provides a highly efficient AAV targeting strategy that is likely to open up new avenues for genetic engineering of cells.
Team – Stahl


Prof. Dr. Felix Stahl, PhD
Research Group Leader






Publications Stahl Lab
Brito LF, Ostermann E, Tödter S, Virdi S, Indenbirken D, Brixel R, Arens R, GrundhoffA, Brune W, Stahl FR. (2026)
Abstract
Differential antiviral T cell immunity in early life impacts the clinical outcome of Cytomegalovirus (CMV) infection, but the underlying mechanisms are not well understood. T cells are known to be involved in protection from CMV disease. To further elucidate these mechanisms, we used a model of respiratory murine CMV (MCMV) infection and adoptive T cell transfers to characterize MCMV-specific T cell responses in early life. We analyzed the effector T cell differentiation using single-cell RNA sequencing and assessed the local pulmonary cytokine milieu. We found delayed enrichment of early-life murine MCMV-specific CD8 T cells due to a general deficiency of αβ T cells. Adoptive transfer of naïve adult T cells into neonates did not protect from lung MCMV infection due to generation of non-cytotoxic CD8 effector T cells. Furthermore, key cytokines required for effective CD8 T cell priming were absent in early life. Supplementation with these cytokines enhanced infection control by transferred adult T cells. The effector function of adult-primed T cells was not disrupted in neonates. Together, this study suggests defective CD8 T cell priming in neonates as a factor explaining the higher risk for MCMV lung disease in the early-life phase.
Brito LF, Ostermann E, Kottlau J, Tödter S, Brixel R, Schüle R, Solano ME, Brune W, Stahl FR. J Virol.
doi:
Abstract
Human cytomegalovirus (HCMV) is the leading infectious cause of congenital disease, but the mechanisms governing vertical transmission remain poorly defined. Murine cytomegalovirus (MCMV) infection in mice is a widely used model to study pathogenesis in vivo. However, the mouse model cannot be used to study congenital infection as viral transmission from mother to fetus in utero does not occur. In this study, we investigated tissue-specific features of the murine materno-fetal barrier that may restrict viral infection. Following high-dose intravenous challenge of wild-type and severely immunocompromised pregnant mice, MCMV replicated efficiently in the maternal liver but infected only a few cells in the placenta, suggesting an intrinsic resistance. Ex vivo analyses of primary placental cells, trophoblast stem cells, and a trophoblast cell line revealed a low susceptibility to MCMV infection compared to other permissive cell types. The resistance of trophoblast cells to MCMV infection correlated with the absence of neuropilin-1 (NRP1), a cellular receptor required for efficient infection of several cell types. Enforced expression of NRP1 in trophoblast cells increased their susceptibility to MCMV infection and replication, suggesting that the resistance of trophoblast cells to MCMV infection is caused by the lack of a critical receptor. These findings further suggest that, in addition to immune-mediated restrictions, cell-intrinsic resistance limits MCMV infection and transmission at the murine materno-fetal interface.
Team – Tolosa


Prof. Dr. rer. nat. Eva Tolosa
Research Group Leader









Publications Tolosa Lab
Bremer SJ, Boxnick A, Glau L, Biermann D, Thiele F, Billeb E, Kolster M, Hackbusch R, Fortmann MI, Tolosa E, Sachweh JS, Gieras A (2024). J of Clin Immunol. 44(3):69.
Abstract
Congenital heart disease (CHD) is the most common birth defect, and up to 50% of infants with CHD require cardiovascular surgery early in life. Current clinical practice often involves thymus resection during cardiac surgery, detrimentally affecting T-cell immunity. However, epidemiological data indicate that CHD patients face an elevated risk for infections and immune-mediated diseases, independent of thymectomy. Hence, we examined whether the cardiac defect impacts thymus function in individuals with CHD. We investigated thymocyte development in 58 infants categorized by CHD complexity. To assess the relationship between CHD complexity and thymic function, we analyzed T-cell development, thymic output, and biomarkers linked to cardiac defects, stress, or inflammation. Patients with highly complex CHD exhibit thymic atrophy, resulting in low frequencies of recent thymic emigrants in peripheral blood, even prior to thymectomy. Elevated plasma cortisol levels were detected in all CHD patients, while high NT-proBNP and IL-6 levels were associated with thymic atrophy. Our findings reveal an association between complex CHD and thymic atrophy, resulting in reduced thymic output. Consequently, thymus preservation during cardiovascular surgery could significantly enhance immune function and the long-term health of CHD patients.
Schneider E*, Winzer R*, Rissiek A, Ricklefs I, Meyer-Schwesinger C, Ricklefs FL, Bauche A, Behrends J, Reimer R, Brenna S, Wasielewski H, Lauten M, Rissiek B, Puig B, Cortesi F, Magnus T, Fliegert R, Müller CE, Gagliani N, Tolosa E. Nat Commun. 2021; 12, 5911-5923.
Abstract
Immune cells at sites of inflammation are continuously activated by local antigens and cytokines, and regulatory mechanisms must be enacted to control inflammation. The stepwise hydrolysis of extracellular ATP by ectonucleotidases CD39 and CD73 generates adenosine, a potent immune suppressor. Here we report that human effector CD8 T cells contribute to adenosine production by releasing CD73-containing extracellular vesicles upon activation. These extracellular vesicles have AMPase activity, and the resulting adenosine mediates immune suppression independently of regulatory T cells. In addition, we show that extracellular vesicles isolated from the synovial fluid of patients with juvenile idiopathic arthritis contribute to T cell suppression in a CD73-dependent manner. Our results suggest that the generation of adenosine upon T cell activation is an intrinsic mechanism of human effector T cells that complements regulatory T cell-mediated suppression in the inflamed tissue. Finally, our data underscore the role of immune cell-derived extracellular vesicles in the control of immune responses.